Research Article
Effect of some Treatments on Seed Health and Viability of Soybean
Department of Seed Technology Research, Field Crops Research Institute, ARC, Giza, Egypt
Soybean (Glycine max L.) is one of the most important oil seed crops in the world. Every population of soybean seeds is a potential carrier of various microorganisms, including fungi, bacteria and viruses (Sinclair and Shurtleff, 1975), also they found that Diaporthe sojae is one of the most important fungi lowering seed quality and affecting germination in soybean, Aspergillus spp., Cercospora kikuchii and Macrophomina phaseolina are the others among fungi contributing to lower seed quality in soybean. Medic-Pap et al. (2007) isolated pathogens fungi genera Diaporthe, Fusarium and facultative parasites, Alternaria, Aspergillus and Penicillium on soybean seeds. Alternaria spp., Cercospora kikuchii, Fusarium spp. Penicillium spp., Aspergillus spp. and Sclerotinia spp., lead to quality problems, deterioration and germination failure of soybean seeds (Gally, 2006). Goulart (2000) detected that F. semitectum, Phomopsis and Cercospora kikuchii with high levels on soybean seed samples. Pioli et al. (1997) isolated pathogens fungi in soybean seeds, Alternaria spp., Cercospora kikuchii, Fusarium spp., Phomopsis spp., Rhizoctonia solani, Curvularia spp. and Nigrospora spp., also they found correlations significant between each pathogen and germination percentage. Phomopsis spp. and Fusarium spp., were predominant in soybean seeds, Also Phomopsis sp., Fusarium spp., Colletotrichum spp., Cercospora spp., Alternaria spp., Aspergillus spp. and Penicillium spp., were identified by Braccini and Dhingra (1996) on soybean seeds. The pathogenic fungal species most frequently observed were Phomopsis spp., F. moniliforme, F. semitectum and Fusarium spp., while found Sclerotinia scleroliorum, Macrophomina phaseolina, Botrytis cinerea and F. oxysporum were detected at low percentages in soybean seed (Conca et al., 1996). Wilson et al. (1995) noticed that fungal damage caused by pathogens of Fusarium, Cercospora and Phomopsis can have a devastating impact on physical quality and seed composition to assess potential utility of highly damaged of soybean where found positive correlation between fungal damage and both protein and oil concen. Aspergillus flavus, A. niger, F. oxysporum and other unidentified fungi were observed abnormal seedlings with high percentage over 25% infection (Singh et al., 1995). Anwar et al. (1995) found that field fungi causing reduction of seed germination or seedling emergence were Alternaria spp., Cercospora kikuchii. Fusarium moniliforme, F. oxysporum, F. solani and M. phaseolina caused root diseases and damping off, while A. alternata, C. kikuchii, Curvularia lunata, Phomopsis sojae and Stemphylium botryosum caused foliage and pod disease. Incidence of Alternaria leaf spot (A. alternate), anthracnose (Colletotrichum dematium), Cercospora leaf spot (C. kikuchii), pod rot and blight (F. semitectum and C. kikuchii) and Rhizoctonia aerial blight (R.. solani) was higher in early sown cultivar compared with late sown, seed treated with thiram gave consistently higher seedling emergence than the untreated seed irrespective of sowing dates (Rahman et al., 1995). Roy and Ratnayake (1997) found that Fusarium semitectum in seeds was negatively correlated with seed germination on agar and with seedling emergence in the greenhouse and reduced root volume, shoot dry weight of seedlings, also they found that seedling emergence was reduced to greater extent poor-quality seeds were inoculated with conidia of F. semitectum compared with high-quality of soybean seeds. Hamman et al. (1996) observed that germination percentage was reduced due to the effect of seed-borne pathogens, which resulted in relatively large numbers of dead seeds, abnormal seedlings and damaged seed coats, also found highly significant correlations between the number of dead seeds and the incidence of fungal infection and between the number of abnormal seedlings, seed coat damage of soybean seeds. Wahid et al. (1995) found that seed treatment soybean with Vitavax gave 100% inhibited growth of F. moniliforme, F. solani and F. oxysporum and improved germination. Seed treatment of soybean with thiram (0.3%) resulted in the highest percentage germination (Charjan and Tarar, 1992). Soybean seeds were treated with carbendaxim+thiram (1+1) at 0.3% eradicated F. oxysporum, F. semitectum, M. phaseolina, A. flavus and A. alternate and increased germination in the filed (Singh, 1997). Sonavane et al. (2011) showed that seed treatment of soybean with fungicidal (thiram+carbendazim) were significantly superior compared to the biological control agents (Trichoderma viride), where lead to lowest seed mycoflora, improved seed germination and seedling vigor index. Saikia et al. (2003) reported that salicylic acid stimulated systemic resistance in chickpea against Fusarium wilt and reduced the disease 23-40%. Morsy et al. (2011) found that Bio-arec (Bacillus megaterium), Bio-zeid (Trichoderma hamatum) and abiotic salicylic acid lead to provide protection against alfalfa downy mildow, rust, root rot and wilt disease when applied as spry treatment or seed soaking. Biotic and abiotic lead to improving seed viability and reduction of fungal diseases as a safe way recommended in controlling of onion seeds (Ibrahim and Kishk, 2014). This study effect of seed coating with fungicide (Vitavax-thiram), seed dressing with biotic (Bio-arec, Bio-zeid) and seed soaking (salicylic acid) on seed, seedling vigor and control of seed-borne fungi on seed and seedling of soybean at Seed Pathology Laboratory, Seed Technology Research Unit, Mansoura, Egypt.
Seed materials: In the present study on samples of soybean from Department of Legumes Crops Research, Field Crops Research Institute, Agriculture Research Center, Egypt. Naturally infected seeds of soybean seven cultivars cvs Giza 21, G.35, G.111, G.22, G.82, Klark, Crawford used for this work in season 2014 for two times. Seed were surface sterilized in 1% sodium hypochlorite solution for 3 min, thin rinsed with sterilized water and air dried.
Seed treatments
Fungicide treatment (seed coating): Soybean seed were used in this experiment for each cultivar were treated with Vitavax-Thiram 200 (carboxin 37.5+thiram 37.5%) at rate 2 g kg–1 seeds and added the polymer coating 10 mL kg–1 were taken in 100 mL dry flasks on a mechanical shaker for about 20 min tell the seeds are uniformly coated, later the treated seeds were spread on a sheet under shade and dried completely. The dried seeds were used for sowing.
Biocides treatment (seed dressing): Two different biological preparations namely Bio-arec (Bacillus megaterium) 25×108 CFU g–1 and Bio-zeid (Trichoderma hamatum or album) 10×108 spore g–1 obtained from Biological Control Unit, Plant Pathology Institute preparation contains active ingredient biocides to the recommendations of the Ministry of Agriculture, Egypt, were used at rate 5 g kg–1 seed of soybean cultivars under study plus 3 mL sterile water, all were mixed properly as seed dressing treatment and air dried for 30 min on sterile tray to enable the seeds to absorb the biocides and used for sowing.
A biotic treatments (seed soaking): Soybean seeds for each cultivar were completely dipped in the solution of salicylic acid concentration 10 mM for 12 h. The treated seeds were dried for 24 h at room temperature and used for sowing.
Seed health evaluation (blotter test): Two hundred of soybean seeds were used in this experiment for each cultivar and treat were plated on three layers of filter paper will soaked in distilled water in eight replicates (25 seeds/Petri dish 12 cm). Other two hundred seeds un treated as a control were incubated at 25°C±2 for seven days under alternating cycles of 12 h near ultraviolet (NUV) light and darkens. Percentage of fungi under investigation were calculated by stereo-binocular microscope (Wild Heerbrugg 6.3-32X) and recorded to the following equation:
![]()
where, n1 the seeds with fungal growth and n2 the number of treated seeds.
Seed and seedling vigor evaluation in pots: One hundred seeds for each treatment and cultivar were planted in 20 cm diameter pots containing sterilized sand in 4 replicates at the rate 25 seeds/pot and other untreated seeds were used as check. Pots were incubated in a growth chamber (Seedburo Equipment Company, USA) for 14 days at 25°C day and night lengths 15/9 h to study the following characters:
| • | Germination percentage (G%) (normal seedlings): It was calculated by counting only normal seedlings (ISTA., 1999) rules: |
![]()
where, MGP is the mean percentage, n1 is number of treated seed plated and n2 the number of abnormal seedlings plus died seeds
| • | Speed Germination Index (SGI): It was calculated as described in the (AOSA., 1983) by the following equation: |
![]()
| • | At the final count, five normal seedlings from each replicate were randomly taken to measure seedling characters, seedling length (cm), dry weight (g) of seedlings (ISTA., 1999) rules: |
Seedling Vigor Index (SVI) = MGB×seedling dry weight
Seedling health evaluation: The seedling test were separated into abnormal seedlings, seed rot and the causal fungi examined using a stereo-scopic microscope and recorded percentage of fungi as described in the technical bulletin on seed-borne diseases (Agarwal et al., 1989; Ellis, 1980):
![]()
Statistical analysis: The observed data was statistically analyzed as the technique of analysis of variance ANOVA of the randomized complete block design as mentioned by Gomez and Gomez (1984). The means were compared using the Least Significant Differences (LSD). Statistical analysis was performed using analysis of variance technique ANOVA by means of MSTAT-C" computer software package.
Identification of soybean seed-borne fungi (seed health evaluation): Seven cultivars of soybean seed samples were tested for using the standard blotter method seedlings (ISTA., 1999) rules, i.e. Giza 21, G. 35, G.111, G. 22, G.82, klark, Crawford. Eleven seed-borne fungi were identified as Alternaria spp., Aspergillus spp., Cercospora kikuchii, Diaporthe sojae, Fusarium moniliforme, F. semitectum, F. solani, F. oxysporum, Macrophomina phaseolina, Rhizoctonia solani and Penicillium spp., associated of soybean seed. Data presented in (Table 1) revealed that the highest frequency percentage was recorded by Aspergillus spp. (7.5%) on cv. G.22, Penicillium spp. (5.6%) on cv. G.82, while C. kikuchii, F. moniliforme and F. solani recorded (3.5%) on cvs G. 22, G. 111, G. 111 and F. oxysporum (3,2%) on cv. Crawford as pathogenic fungi, also cv. G. 22 was the highest of the total fungi (28.4) followed by cvs. G. 35, G. 82, G. 111, Clark, Crawford and G. 21 was the less (18.4).
Also, data presented in Table 1 indicated that Vitavax-200 used for seed coating lead to eliminate of most fungi, meanwhile Bio-arec as seed coating reduced Aspergillus spp., from 13.7-4%, C. kikuchii from 4.4-1.1%, D. sojae from 4.2-0.6% and F. moniliforme from 8-1.5%, Also, Bio-zeid was able to reduced F. oxysporum from 5.9-1.3%, M. phaseolina from 1.7-0%. Salicylic acid as seed soaking showed least of fungal infection percentage such as Penicillium spp., from 12-4.9%, F. solani from 3.8-0%, F. semitectum from 6.5-1.2%, F. moniliforme from 8-0.4% and total fungi from 67-13. Generally, effect all treatments were high significant with fungal infection percentage for each fungi.
Interaction between cultivars, treatments and the effect on seed-borne fungi associated of soybean seed (Table 2), indicated that on seed of cv. G. 21, Aspergillus spp., recorded the highest frequency percentage (16%) followed by Penicillium spp. (9.3%), D. sojae (5.3%), F. oxysporum (5.3%) as control.
| Table 1: | Frequency of occurrence of seed-borne fungi associated with cvs soybean after seed treatment by Blotter method |
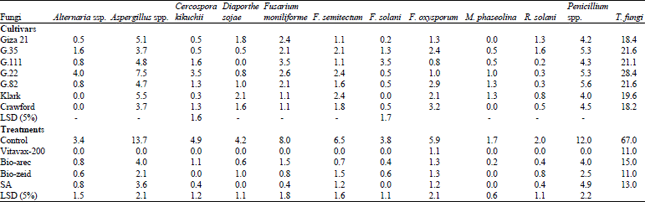 | |
| Test was carried out using blotter technique, Two hundred seeds were tested, Incubation was carried out at 25°C±for 7 days (ISTA., 1999), SA: Salicylic acid, LSD: Least significant difference | |
Cultivar G. 35 revealed that Penicillium spp. was (13.3%) followed by Aspergillus spp. (12%) thin F. moniliforme, F. semitectum (6.6%) and F. solani (5.3%). Fusarium solani was 13% and both Aspergillus spp. and F. moniliforme were (12%), while C. kikuchii and F. semitectum (5.3%) on cv. G. 111as control. On cv. G. 22 recorded by blotter test, Aspergillus spp. and Penicillium spp. 24 and 14.6%, respectively, C. kikuchii was (13.3%) and F. semitectum (8%). Aspergillus spp., was (13%), Penicillium spp. (14.6%), F. oxysporum (9.3%), F. moniliforme (8%), F. semitectum (6.7%) and C. kikuchii (5.3%) on seed of cv. G. 82. Also, results pointed out that Aspergillus spp. and Penicillium were the highest infection percentage (12%) followed by F. semitectum (9.3%), while D. sojae and F. oxysporum were (6.6%) on seed of cv. Klark. Crawford cultivar recorded with Penicillium spp. (12%), F. oxysporum 10.6 and 6.6% both D. sojae and Aspergillus spp., respectively, F. moniliforme and F. semitectum were (5.3%). On the other hand, cv. G. 22 recorded the highest number of total fungi (98.6) followed by cv. G. 82 (68.8), cv. G111 (68.4), cv. G. 35 (62.3), Klark (57.2), Crawford (55.7) and cv. G. 21 (54.4) as control. Data in Table 2 indicated that the fungicide (Vitavax-200) was effective in almost, eliminated of seed-borne fungi to all cultivars used in this experiment, while biotic (Bio-arec) reduced total fungi from 98.6-26.4, (Bio-zeid) to 14.6, a biotic (SA) reduced total fungi to 5.3 on seed of cv. G. 22. Also, results showed that Penicillium spp. recorded the highest number of the total fungi (167.1) and the more frequency on cvs soybean seed as survey compared with other fungi, followed by Aspergillus spp. (163.3) thin F. moniliforme (74.1), F. oxysporum (68.5), F. semitectum (63.3), C. kikuchii (44.9), Alternaria spp. (42.5), D. sojai (39.6), F. solani (32.8), R. solani (25) and M. phaseolina (14.1).
Effect of cultivars and seed treatments under study on seed and seedling vigor of soybean Table 3 shows that cv. Giza 82 recording the highest value of Mean Germination Percentage (MGP) (88%), Speed Germination Index (SGI) (8.4) and Seedling Length (SL) (24.5 cm), while cv. G. 21 was the less value of MGP (80%), SL (18.5 cm) and dry weight (DG) (1.2 Gm.) as compared cultivars. On the other hand, effect of treatments were high significant to characters vigor, seed soaking with Salicylic Acid (SA) showed better germination (MGP) (91%), SPI (8.6), SL (30 cm), DW (1.7 Gm.) and gave the highest value of seedling vigor index followed by Vitavax-200 gave (89%), MGP, Bio-zeid (88%) MGP and Bio-arec (87%) MGP as compared with control (68%).
Seedling health evaluation, abnormal seedlings and seed rot infection with pathogenic fungi tabulated in Table 4. Abnormal seedlings recorded the highest Percentage of Fungal Infection (PFI) by D. sojae, F. oxysporum and R. solani were (9%) to cvs Klark, G. 35 and G. 111 as control, respectively, while F. solani recorded 7 and 8% with cv. G. 111 and Klark, respectively. Fusarium semitectum gave percentage of fungal infection (5%) with cvs G. 35 and G. 21, while C. kikuchii recorded (4%) with cvs G. 21, G. 35 and G. 82. Macrophomina phaseolina gave PFI with abnormal seedlings of cvs G. 111, Klark and G. 21 5, 4 and 3%, respectively as control. Seed rot type gave to F. semitectum and F. moniliforme the highest value (5%) with cv. G. 111 and (4%) with cv. Klark, respectively. Generally, on seed of cvs soybean found lowering of MGP due to association high percentages of pathogenic fungi for seed and seedlings as C. kikuchii, D. sojae, F. moniliforme, F. semitectum, F. solani, F. oxysporum, M. phaseolina and R. solani.
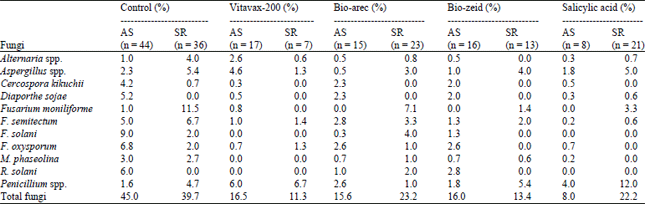
Table 5 showed that the effect of treatments on percentage of fungi and relation to abnormal seedlings and seed rot, F. solani was the more fungi associated of abnormal seedlings with (9%) followed by F. oxysporum (6.8%) thin R. solani (6%), D. sojae (5.2%), F. semitectum (5%), C. kikuchii (4.2%) and M. phaseolina (3%) as control, respectively (Table 5). Also F. moniliforme has been widely and most frequently with seed rot where recorded (11.5%) followed by F. semitectum thin Aspergillus spp. and Penicillium spp.
| Table 2: | Interaction between cultivars, treatments and the effect on seed borne fungi associated of soybean seed |
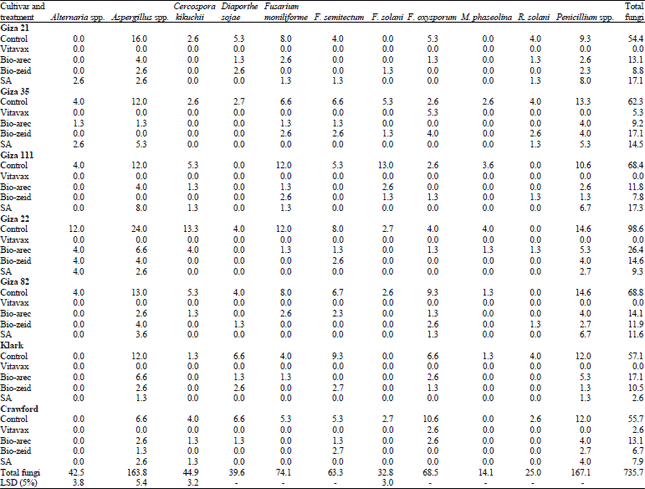 | |
| Table 3: | Effect of cultivars and seed treatments on seed and seedling vigor of soybean in Pots |
 | |
| G (%): Mean germination percentage, SG: Speed germination, SL: Seedling length, AS: Abnormal seedlings, SR: Seed rot, DW: Dry weight, SVI: Seedling vigor index, LSD: Least significant difference | |
On the other hand Vitavax-200 lead to elimination fungi such as C. kikuchii, D. sojae, F. moniliforme, F. solani, M. phaseolina and R. solani in seed rot, Bio-arec lead to less number of infection by pathogenic fungi compared to the control as F. oxysporum from 6.8-2.6% and R. solani from 6-1% in abnormal seedlings. Bio-zeid less seed infection of F. moniliforme from 11.5-1.4%, also F. semitectum from 6.7- 2% with seed rot. Salicylic acid lead to eliminating and/or reducing number of pathogenic fungi on both abnormal seedlings and seed rot as compared with control and other treatments.
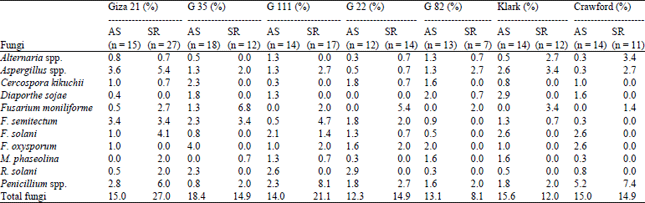
Table 6 on seed of Giza 21 show that F. semitectum caused the highest infection percentage of abnormal seedlings (3.4%), F. solani (4.1%) on seed rot. Also, on cv. G. 35 recorded F. moniliforme the highest infection on seed rot (6.8%) followed by G. 22 (5.4%), while C. sojae was (2.3%) on abnormal seedlings with cv.G.35. Giza 82 cv. was the less cultivars effective with total fungi on abnormal seedlings and seed rot as compared other cultivars, on the other hand cv. G. 21 was the more cultivars as seed-borne of total fungi in abnormal seedlings and seed rot.
| Table 4: | Percentage of fungal infection in abnormal seedlings and seed rot after seed treatment of soybean cultivars in pots |
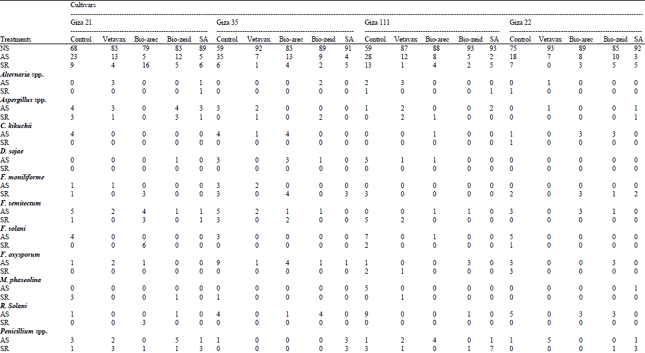 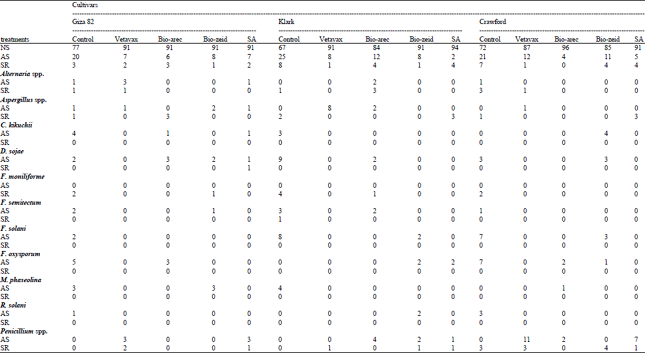 | |
| NS: Normal seedlings, plumlee and root well developed, AS: Abnormal seedlings, decay in shoot and root, primary root showing browning no branching or secondary development shoot usually appearing weak, SR: Seed rot, consisted of non-germinated seeds covered with hyphal growth of the tested (Guerrero et al.,1972) | |
| Table 5: | Effect of treatments on percentage of fungal infection and relation to percentage of abnormal seedlings and seed rot of soybean |
 | |
| Table 6: | Percentage of fungal infection and relation to abnormal seedlings, seed rot and cultivars of soybean |
 | |
Seed-borne fungal inocula are known to cause several levels of lowering seed quality and affecting germination in soybean (Sinclair and Shurtleff, 1975). The present study investigated 7 cultivars of soybean seed by blotter method and identification 11 species 8 genera, namely Alternaria spp., Aspergillus spp., C. kikuchii, D. sojae, F. moniliforme, F. semitectum, F. solani, F. oxysporum, M. phaseolina, R. solani and Penicillium spp. (Medic-Pap et al., 2007; Goulart, 2000; Pioli et al., 1997). These results point to Aspergillus spp., was the more frequency fungi on seeds followed by Penicillium spp., both it from saprophyte fungi but found that cases low percentages in normal seedlings and deterioration in vigor seed, Krishnamurty and Raveesha (1996) indicated that Aspergillus values was the frequent occurrence of orange-yellow discolored which were highly distorted, shriveled and smaller than normal seeds and isolated from seed coats, cotyledons and internal tissues of the discolored seeds. On the other hand, data obtained showed that C. kikuchii, D. sojae, Fusarium spp., M. phaseolina and R. solani as pathogenic fungi were observed on abnormal seedlings and seed rot with high percentage and causing reduction of seed germination or seedling emergence. These results were in harmony with those reported by Anwar et al. (1995), Singh et al. (1995), Roy and Ratnayake (1997) and Hamman et al. (1996). Fusarium spp., observed associated with seed rot and abnormal seedlings and produce rotten lesions on cotyledons and hypocotyls also plumlee soft rot of soybean seedlings (Gally et al., 1998). Fusarium oxysporum externally appeared shrunken, slightly irregular in shape, often with cracks in the seed coat with light to dark pink discolored areas over most of the infected seed surface, seeds with severe symptoms did not germinate, hypha were found spread over the surface of the seed coat producing macro-and micro conidia and within the hilum region (Velicheti and Sinclair,1991). Fusarium wilt caused by F. oxysporum is one of the major diseases of soybean, the fungus invades plant vascular tissues and induces sever wilting of the foliage by blocking xylem transport and impeding the movement of water, the pathogen is both seed and soil borne. Haikal (2008) soaked seeds of soybean with culture filtrates detected that percentage seed germination and seedling growth decreased with increase in filtrate concentration, filtrate age and pre-soaking time in all the fungal filtrates A. niger, F. culmorium, Penicillium spp. and R. solani. Macrophomina phaseolina as seed transmission causing charcoal rot, grey to black fungal growth were observed on the seed plate by blotter method or agar plate method, while under laboratory and green house conditions resulted seedling mortality, seed rot and reducing germination indicating the transmission of M. phaseolina from infected seeds to seedlings (Mandhare et al., 2009). Phomopsis spp. (Diaporthe sojae) are associated with non germinated seeds, increase of seed infection by seed-borne Phomopsis spp. and other fungi seems to reduce the germination of soybean seeds (Arulnandhy, 1983). The obtained results showed that fungicidal treatments Vitavax-200 with recommended doses eliminated the fungus and improved seed germination, increased normal seedlings, decreased abnormal seedlings and seed rot of soybean cultivars under experiment conditions, earlier studies by Charjan and Tartar (1992), Singh (1997) and Sonavane et al. (2011). Seed treatment with 2.5 g. captan/1 kg. seed and 2.5 g. thiram/1 kg seed against Diaporthe sojae, A. alternate, A. flavus, Curvularia lunata and F. oxysporum improved seed germination, seedling length and dry weight (Manshi et al., 2004). Solanke et al. (1997) detected that thiram improving the germination percentage, controlled pre- and post-emergence mortality caused by Aspergillus spp., F. moniliforme, Curvularia lunata, A. alternate and Penicillium spp. Mean of control showed that using treatment as seed dressing of biocides (Bio-arec and Bio-zeid) reduced percentage of mean infection seed-borne fungi under study and enhanced for characters vigor of seed and seedlings soybean, these results agree with those reported by Sonavane et al. (2011) and Saikia et al. (2003). Also, Sonavane et al. (2011) found that Aspergillus spp., infection resulted in the lowest seed germination and seedling vigor index followed by F. oxysporum, F. moniliforme thin M. phaseolina and treatments with fungicidal (thiram+carbendazim) were significantly compared to the biological control agents (Trichoderma viride). Biological control of plant diseases can occur through different mechanisms, which are generally classified as: antibiosis, competition, suppression, direct parasitism, induced resistance, hypo virulence and predation (Moyer and Peres, 2008).
Present results showed that seed soaking of soybean with Salicylic acid have positive effect on reduced percentage of mean infection seed-borne fungi, number of total fungi, abnormal seedlings and seed rot besides enhancing in growth parameters such as Mean Germination Percentage (MPG), Speed Germination (SG), Seedling Length (SL) and Dry Weight (DW) compared with control, similar reasons were reported by Morsy et al. (2011) and Ibrahim and Kishk (2014). The mode of action of abiotic inducers for controlling plant diseases may include: (1) Acting as second messengers in enhancing the host defense mechanism (Geetha and Shetty, 2002), (2) Activating resistance by increasing the activity of peroxidase (Hassan et al., 2007), (3) Activating resistance through inhibition of some antioxidant enzymes and catalases (Radwan et al., 2008) and (4) Enhancing resistance by direct effects on multiplication, development and survival of pathogens or indirect effects on plan metabolism, with subsequent effects on the pathogen food supply (Khan et al., 2003). Exogenous application of SA may influence stomata closure ion uptake and transport inhibition of ethylene biosynthesis, transpiration and stress tolerance (Khan et al., 2003).
Generally, data obtained through this investigation showed that treatment with seed coating (Vitavax-thiram200) gave improving characters of seed and seedling vigor and eliminated or reduced numbers of pathogenic fungi, biotic and abiotic (seed dressing and soaking) inducers can be safely recommended in controlling fungal diseases and enhancing of seed and seedling vigor of soybean.
V. Bharathi Reply
Sir
I want the full length of your paper and please send me the paper to this mail id as early as possible.its a good paper and i want this paper. Sir I want my other papers to publish in this journal. I request to kindly help me in this regard.