ABSTRACT
This study was conducted to evaluate Iraqi propolis against gray mold on orange. Propolis Ethanolic Extract (PEE) in three concentrations 1, 2 and 3% were used to treated orange treatments separately and storage at 25±2°C for three weeks. Results showed a significant reduced in disease severity in the first, second and third week for all PEE concentrations compared with infected control. The best treatment was 3% in disease severity reduction. Disease incidence was significantly reduced with the treatment treated with PEE for all concentrations compared with the pathogen treatment, both concentration of PEE 2 and 3% was the most effective. Also, all PEE concentrations reduce depth of the mold area compared with pathogen treatment. There are no significant differences in patulin production between PEE and pathogen treatments was founded.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ppj.2015.153.157
URL: https://scialert.net/abstract/?doi=ppj.2015.153.157
INTRODUCTION
Green mold caused by Penicillium digitatum is one of major necrotroph fungal disease caused serious damage to citrus fruit (Janisiewicz and Korsten, 2002; Brown et al., 2000). Fungal and pest decay caused up to half of all fruits harvested is lost in worldwide (Burden et al., 1989). These diseases could cause a loss of up to 10-30% decrease in crop yield and marketing quality (Agrios, 2005; Serrano et al., 2005). According to FAO in 2012, Citrus is an important fruit crop in world production estimated at 115 million tone per year during 2010-2011.
Plant extract used as alternative control to control plant disease, to replace the conventional synthetic pesticides (Viuda-Martos et al., 2007; Al-Samarrai et al., 2013). In the recent years propolis has been used as a health supplement suited to consumers, propolis a natural honey bee product have biological activities. It is a resinous substance collected by Apis mellifera L. from various tree buds.
Ethanolic extract of Turkish propolis (PEE) collected from various areas show multi-properties activity as antibacterial (Keskin et al., 2001; Ozcan et al., 2004), antifungal (Koc et al., 2005), antioxidant (Kolankaya et al., 2002; Orhan et al., 1999) and anticarcinogenic (Ozkul et al., 2005). The chemicals compounds in propolis and biological activities depend on different factors such as the geographical regions, collection time and plant source (Bankova, 2005; Hegazi et al., 2014). The propolis components is variable depends on many factors, its approximately 50% resin, 30% wax, 10% essential oils, 5% pollen and 5% other organic compounds (Falcao et al., 2010). A few studies have been made to test the antimicrobial activity of propolis in foods. Han et al. (2000) reported that propolis extracts have a good chemical that used to preservatives pork meat products.
The propolis efficacy was used to extending fruits life storage and preventing fungal decay in different fruits during storage conditions, has been improved by several studies (Candir et al., 2009; Ozdemir et al., 2010). Candir et al. (2009) found that storage sweet Cherry, treated by dipping in 5% of PEE, prevent fungal decay for four week at 0°C. Ozdemir et al. (2010) also found that dipped Star Ruby grapefruit in PEE at 5% concentrations was effective to prevent Penicillium spp. fungal decay. Under cold storage conditions, treated the orange fruit with 3% PEE found to be the most effective concentration to reduced fruit decay that caused by Penicillium spp. (El-Badawy et al., 2012).
Another study found that PEE at 10% concentration showed 100% inhibition of mycelial growth of Penicillium spp. on PDA (Ayhan et al., 2013). Propolis was used as an antimicrobial against spoiled fruit juices, to preserved nonpasteurized fruit juice (Koc et al., 2007). A previous study found that treated with 5% of PEE prevent snap bean rotten with white mold Sclerotinia sclerotiorum in storage conditions (Matny et al., 2014). A molecular study of the P. digitatum genome showed that patulin can be produce by this species (Marcet-Houben et al., 2012). This study aims to use PEE to improved and give a long shelf-life storage for orange fruit.
MATERIALS AND METHODS
Pathogen isolation: The pathogen P. digitatum were isolated from decayed oranges fruits collected from different market places in Baghdad, direct isolated of the pathogen by taking direct smear of spores from the surface of the fruit and placed on petridish plates contained PDA medium. Petridishes incubated at 25±2°C for 5 days.
Pathogenicity test: Ten isolates of P. digitatum were tested on orange fruit to estimate the most aggressive isolate to use it in the subsequent experiment. Three oranges fruit for each isolate were surface sterilized by dipping in 10% sodium hypochlorite for 5 min and washed two times with sterilized water. The fruit were surface wounded with cork porer 0.5 cm, 23×105 suspension spores was prepared for each isolate and 100 μL was used as inoculation for each fruit. The inoculated fruit were kept in plastic box 20×10×10 cm and incubated at 25±2°C for 10 days. The most aggressive isolate was chosen for the subsequent test.
Propolis Ethanolic Extract (PEE) preparation: Propolis were collected from bee hives in Baghdad. Two hundred gram of propolis were kept in freezer for 3 h and grounded directly by coffee grinder, 69% ethanol was used for extraction in ratio 1:3 propolis:ethanol and kept on shaker 150 rpm for 2 days. The extract were filtrated by using centrifugation at 5000 rpm for 10 min, supernatant were collected and evaporated at room temperature (25°C) for 3 days. The remind resin were collected to use in subsequent test.
Antifungal activity test: The PEE extracts were added into PDA media after dissolve the propolis resin with a small amount of ethanol and complete the volume with water. Three consternation of 0.5, 1.5 and 3% were prepared. A disc of 0.5 cm diameter of fungal culture on PDA of 7 days old was placed at the center of each petridishes and incubated at 25±2°C (3 replication for each concentration). The inhibition of fungal growth was calculated as following:
where, dc = average diameter of linear growth in control. dt = average diameter of linear growth in treatment.
Storage experiment: Three concentrations of propolis extract were prepared, 1, 2, 3% of propolis resin weight and dissolved in 10 mL of 96% ethanol, the volume completed with sterile distilled water to 100 mL. Seventy orange fruit were surface sterilized with 10% sodium hypochlorite for 15 min and washed two time with sterilized water. After a surface dried of fruit they were surface wounded of 0.5 cm by using crock porer. Three orange fruit were soaked in the prepared PEE in each concentration separately for 1 min, the fruit kept in lab temperature entail dried, three orange fruit were socked in sterilized water as non-inoculated control treatment and three orange fruit were inoculated with the pathogen without treatment with PEE as inoculated control. Three replicate for each treatment were conducted. One Hundred microliter of 23×105 spore suspensions of P. digitatum add to each fruit in the wounded holes.
All treatment were placed on plastic box 20×10×10 cm and kept on 25±2°C until control treatment (pathogen inoculated) completely molded. Disease incidence and disease severity were calculated depending on:
This disease scale used for disease severity: The 0 = no infection, 1= quarter of orange fruit decay, 2 = half of orange fruit decay, 3 = three quarters orange fruit decay, 4 = all orange fruit decay:
The depth of the molded area was measured also. All experiment were CRD design, All treatments were placed in control temperature room, the data was statically analysis by using Gen Stat program.
Patulin extraction: Patulin ELISA test kit from Glory Science Co. Ltd (USA) were used. Each orange fruit were blending with 100 mL distilled water. Ten milliliter of orange juice were transfer to separate funnel, 20 mL of acetonitrile with shaking for 1 min. Carefully take 100 μL of the supernatant and mix with 300 μL of 1 X patulin dilution buffer, 100 μL from each sample were used to measured patulin. The patulin concentrations were measured by draw standard carve and toxin concentration were calculated (company kit protocol).
RESULTS
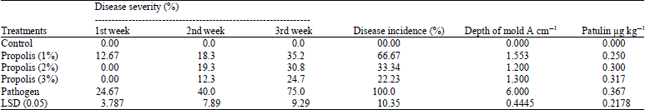
Results showed in the first week significant effective of PEE in all concentrations to reduce disease severity, it was 12.62, 0.00 and 0.00% for PEE 1, 2 and 3%, respectively, compared with pathogen treatment 24.67%. The second week results showed significant decrease in disease percentage in PEE treatments compared with pathogen treatment only and there are no significant difference between PEE treatments, it was 18.3, 19.3, 12.3 and 40.0%, respectively.
| Table 1: | Effect of PEE extract on gray mold disease severity, depth of mold area and patulin production on oranges fruit infected by P. digitatum |
 | |
 | |
| Fig. 1(a-e): | Efficacy of PEE on storage orange to prevent gray mold, (a) 1% PEE, (b) 2% PEE (c) 3% PEE, (d) Control non inoculated with pathogen and (e) Pathogen only |
Third week results of PEE treatments found to reduce the disease severity percentage compared with pathogen treatment, it was 35.2, 30.8, 24.7 and 75.0%, respectively. The PEE 3%, was the best treatment that give the significantly lowest disease severity compared with 1 and 2% of PEE treatments it was 24.7, 35.2 and 30.8%, respectively. All PEE treatments showed a significant decrease in the percentage of disease incidence compared with pathogen treatment. The treatment 2 and 3% PEE showed significant reduce in disease incidence in compared with 1% PEE, it was 33.34, 22.23 and 66.67%, respectively.
The depth of molded area in infected orang’s fruit treatment that treated with PEE showed the less depth of molded area compared with pathogen treatment only, it was 1.553, 1.200, 1.300 and 6.000 cm of 1, 2 and 3% PEE and pathogen treatment, respectively.
The effect of PEE on patulin production in infected oranges fruit were study. Result showed that there are no significant efficacy of PEE to reduce patulin production in all treatments compared with pathogen treatments, it was 0.250, 0.300, 0.317 and 0.367 μg kg‾1 for PEE 1, 2, 3% and pathogen treatment, respectively (Fig. 1 and Table 1).
DISCUSSIONS
The goal of this study is to evaluate PEE to control orange gray decay disease caused by P. digitatum under storage conditions. P. digitatum known as pathogen caused a serious damage and losses more than 30% in post-harvest and marketing conditions (Agrios, 2005).
Several study exam the antimicrobial activity of PEE in vitro and in vivo conductions, the different sources of propolis collecting by the bees have various effect as antimicrobial efficacy. The PEE contains many of antifungal compounds that have activity against fungi that’s may be inhibition mycelium grow and spore germination. Many studies have mention about the active ingredient of PEE against fungi, Peng et al. (2012) that pinocembrin bioactive compound isolated from propolis have strong antifungal activity against P. italicum that caused decay on orange, the suggest that efficacy of pinocembrin its maybe due to interfering energy homeostasis and cell membrane damage. Mello et al. (2006) suggested that the antifungal activity of propolis maybe due to the changes in the cell wall that leading to increasing of volume and membrane rupture. Previous study found three compound isocupressic acid, (+)-agathadiol and epi-13-torulosol that have antifungal activity against the tested fungi on culture medium by reduce radial mycelium grow (Meneses et al., 2009). Ozcan et al. (2003) also reported that the methanolic propolis extract had large scale effect to inhibition mycelial growth against many phytopathogenic fungi in culture medium. Propolis also have many active compound like aromatic acid, phenolic and polyphenolic compound and triterpenes etc that have antimicrobial properties against fungal (Burdock, 1998). Silici and Karaman (2014) found activity of Turkish propolis to reduce patulin production in apple juice, the mode of action of prevent patulin it maybe due to direct effect of fungal metabolism that lead to inhibition toxin production, or inhibition of mycelia growth that led to prevent patulin production.
REFERENCES
- Bankova, V., 2005. Recent trends and important developments in propolis research. Evidence-Based Complement. Altern. Med., 2: 29-32.
CrossRefPubMedDirect Link - Brown, G.E., C. Davis and M. Chambers, 2000. Control of citrus green mold with Aspire is impacted by the type of injury. Postharvest Biol. Technol., 18: 57-65.
CrossRefDirect Link - Burdock, G.A., 1998. Review of the biological properties and toxicity of bee propolis (propolis). Food Chem. Toxicol., 36: 347-363.
CrossRefPubMedDirect Link - Candir, E.E., A.E. Ozdemir, E.M. Soylu, N. Sahinler and A. Gul, 2009. Effects of propolis on storage of sweet cherry cultivar Aksehir Napolyon. Asian J. Chem., 21: 2659-2666.
Direct Link - Falcao, S.I., M. Vilas-Boas, L.M. Estevinho, C. Barros, M.R.M. Domingues and S.M. Cardoso, 2010. Phenolic characterization of Northeast Portuguese propolis: Usual and unusual compounds. Anal. Bioanal. Chem., 396: 887-897.
CrossRefDirect Link - Al-Samarrai, G.F., H. Singh and M. Syarhabil, 2013. Extracts some plants on controlling green mold of orange and on postharvest quality parameters. World Applied Sci. J., 22: 564-570.
Direct Link - Han, S.K., K. Yamauchi and H.K. Park, 2000. Effect of nitrite and propolis preservative on volatile basic nitrogen changes in meat products. Microbios, 105: 71-75.
Direct Link - Hegazi, A., A.M. Abdou and F. Abd-Allah, 2014. Egyptian propolis 11: Its antimicrobial activity with comparison with different localities. Int. J. Curr. Microbiol. Applied Sci., 3: 530-538.
Direct Link - Janisiewicz, J.W. and L. Korsten, 2002. Biological control of postharvest diseases of fruits. Annu. Rev. Phytopathol., 40: 411-441.
CrossRefDirect Link - Keskin, N., S. Hazir, S.H. Baser and M. Kurkcuoglu, 2001. Antibacterial activity and chemical composition of Turkish propolis. Z. Naturforsch., 56c: 1112-1115.
Direct Link - Koc, A.N., S. Silici, D. Ayangil, A. Ferahbas and S. Cankaya, 2005. Comparison of in vitro activities of antifungal drugs and ethanolic extract of propolis against Trichophyton rubrum and T. mentagrophytes by using a microdilution assay. Mycoses, 48: 205-210.
PubMed - Kolankaya, D., G. Selmanoglu, K. Sorkun and B. Salih, 2002. Protective effects of Turkish propolis on Alcohol-induced serum lipid changes and liver injury in male rats. Food Chem., 78: 213-217.
CrossRefDirect Link - Marcet-Houben, M., A.R. Ballester, B. de la Fuente, E. Harries, J.F. Marcos, L. Gonzalez-Candelas and T. Gabaldon, 2012. Genome sequence of the necrotrophic fungus Penicillium digitatum, the main postharvest pathogen of citrus. BMC Genomics, Vol. 13.
CrossRef - Mello, A., R.T. Gomes, S. Lara, G. Silva and B. Alves et al., 2006. The effect of Brazilian propolis on the germ tube formation and cell wall of Candida albicans. Pharmacologyonline, 3: 352-358.
Direct Link - Meneses, E., D.L. Durango and C.M. Garcia, 2009. Antifungal activity against postharvest fungi by extracts from colombian propolis. Quimica Nova, 32: 2011-2017.
CrossRefDirect Link - Orhan, H., S. Marol, I.F. Hepsen and G. Sahin, 1999. Effects of some probable antioxidants on selenite-induced cataract formation and oxidative stress-related parameters in rats. Toxicology, 139: 219-232.
CrossRef - Ozcan, M., A. Unver, D.A. Ceylan and R. Yetisir, 2004. Inhibitory effect of pollen and propolis extracts. Nahrung, 48: 188-194.
CrossRef - Ozkul, Y., S. Silici and E. Eroglu, 2005. The anticarcinogenic effect of propolis in human lymphocytes culture. Phytomedicine, 12: 742-747.
CrossRefPubMedDirect Link - Peng, L., S. Yang, Y.J. Cheng, F. Chen, S. Pan and G. Fan, 2012. Antifungal activity and action mode of pinocembrin from propolis against Penicillium italicum. Food Sci. Biotechnol., 21: 1533-1539.
CrossRefDirect Link - Serrano, M., D. Martinez-Romero, S. Castillo, F. Guillen and D. Valero, 2005. The use of natural antifungal compounds improves the beneficial effect of MAP in sweet cherry storage. Innovative Food Sci. Emerging Technol. 6: 115-123.
CrossRefDirect Link - Silici, S. and K. Karaman, 2014. Inhibitory effect of propolis on patulin production of Penicillium expansum in apple juice. J. Food Process. Preserv., 38: 1129-1134.
CrossRefDirect Link - Ayhan, T., A.S. Mumcu, A.O. Tuylu, K. Sorkun and B. Salih, 2013. Antifungal activity of propolis samples collected from different geographical regions of turkey against two Food-related molds, Aspergillus versicolor and Penicillium aurantiogriseum. GIDA, 38: 135-142.
Direct Link - Viuda-Martos, M., Y. Ruiz-Navajas, J. Fernandez-Lopez and J.A. Perez-Alvarez, 2007. Antifungal activities of thyme, clove and oregano essential oils. J. Food Saf., 27: 91-101.
CrossRef








