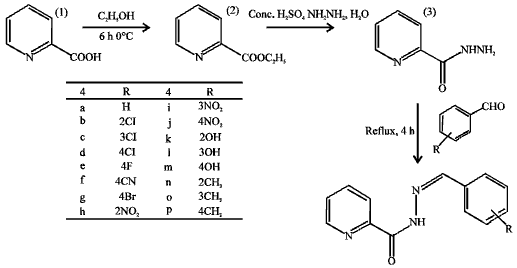
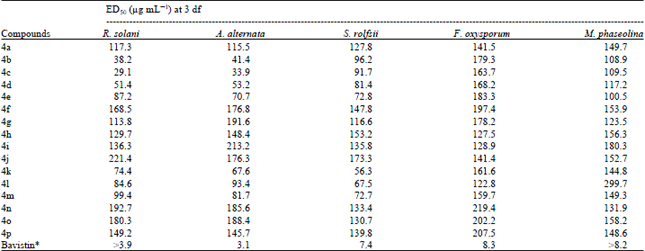
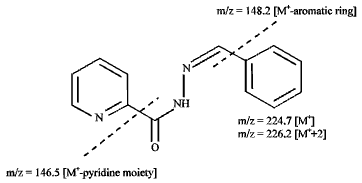
Research Article
In vitro Antifungal Activity of Novel Picolinamides against Soil Borne Fungi and Structure Activity Relationship
Division of Agricultural Chemicals, Indian Agricultural Research Institute, Delhi, 110012, India
Supradip Saha
Division of Agricultural Chemicals, Indian Agricultural Research Institute, Delhi, 110012, India