Research Article
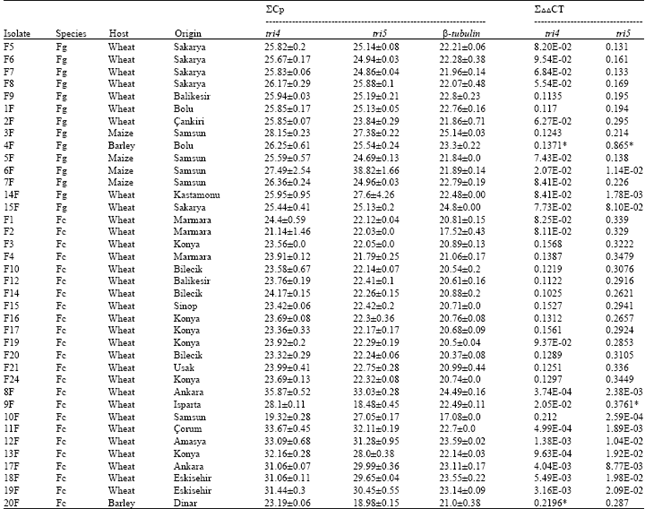
Tri4 and tri5 Gene Expression Analysis in Fusarium graminearum and F. culmorum Isolates by qPCR
Programme of Molecular Biology and Genetics, Institute of Science, Istanbul University, Vezneciler, Istanbul, Turkey
Gulruh Albayrak
Department of Molecular Biology and Genetics, Faculty of Science, Vezneciler, Istanbul University, Istanbul, Turkey