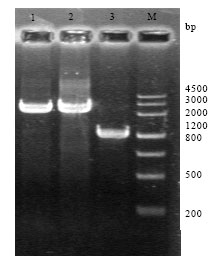
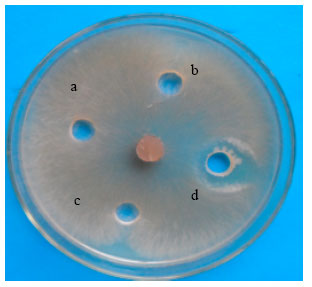
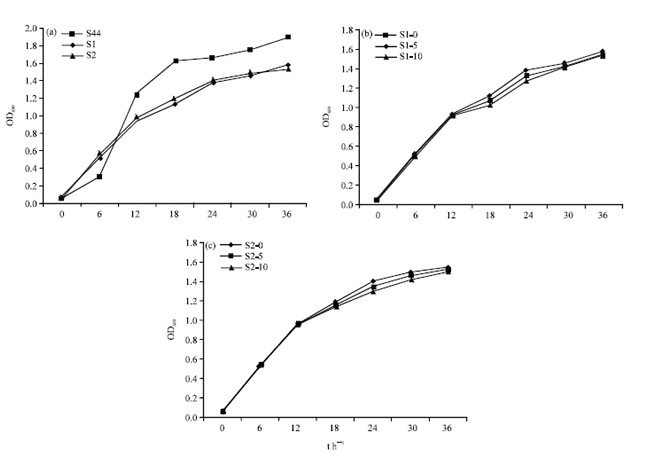
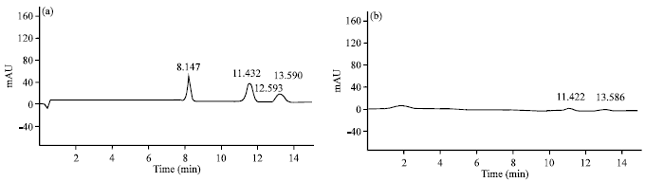
Research Article
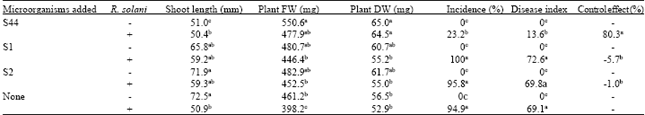
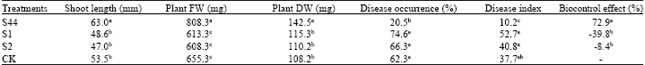
Knockout of ituD Gene of Bacillus subtilis S44 Strain and Impact of its Biocontrol Effect to Cotton Rhizoctoniosis
Key Laboratory at Universities of Xinjiang, Uygur Autonomous Region for Oasis,Agricultural Pest Management and Plant Protection Resource Utilization,Shihezi University, Shihezi, 832003, China
Li Chen
Key Laboratory at Universities of Xinjiang, Uygur Autonomous Region for Oasis,Agricultural Pest Management and Plant Protection Resource Utilization,Shihezi University, Shihezi, 832003, China
SiFeng Zhao
Key Laboratory at Universities of Xinjiang, Uygur Autonomous Region for Oasis,Agricultural Pest Management and Plant Protection Resource Utilization,Shihezi University, Shihezi, 832003, China
Juan Ren
Key Laboratory at Universities of Xinjiang, Uygur Autonomous Region for Oasis,Agricultural Pest Management and Plant Protection Resource Utilization,Shihezi University, Shihezi, 832003, China
XiaoLei Cao
Key Laboratory at Universities of Xinjiang, Uygur Autonomous Region for Oasis,Agricultural Pest Management and Plant Protection Resource Utilization,Shihezi University, Shihezi, 832003, China