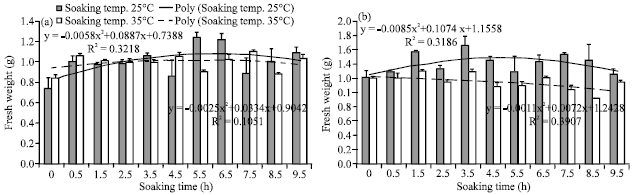
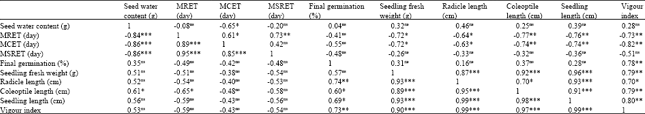
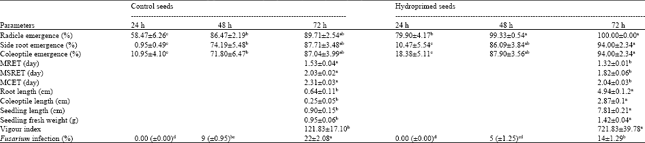
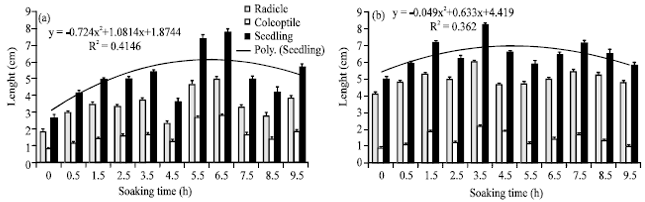
Research Article
Seed Hydropriming Effect on Triticum durum and Hordeum vulgare Germination, Seedling Growth and Resistance to Fusarium culmorum
Laboratory of Molecular Physiology of Plants, Centre of Biotechnology of Borj Cedria, B.P. 901, Hammam-Lif 2050, Tunisia