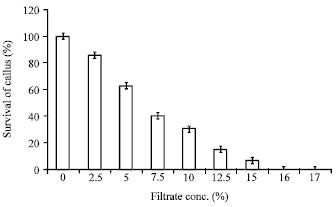
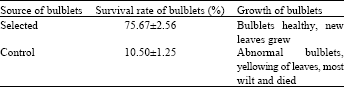
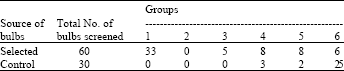Research Article
Development of Mutant Lines of Oriental Hybrid Lily Resistant to Fusarium oxysporum f.sp. lilii Toxin(s)
Department of Biotechnology, University of Horticulture and Forestry, Solan-173230, Himachal Pradesh, India
Surinder Kumar
Department of Biotechnology, University of Horticulture and Forestry, Solan-173230, Himachal Pradesh, India
Minerva Ghani
Department of Biotechnology, University of Horticulture and Forestry, Solan-173230, Himachal Pradesh, India
Manisha Thakur
Department of Biotechnology, University of Horticulture and Forestry, Solan-173230, Himachal Pradesh, India
Harender Raj
Department of Mycology and Plant Pathology, University of Horticulture and Forestry, Solan-173 230, Himachal Pradesh, India