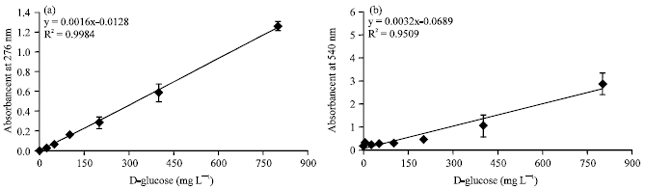
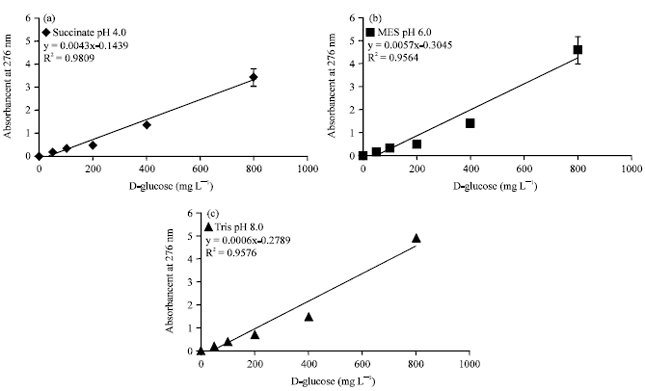
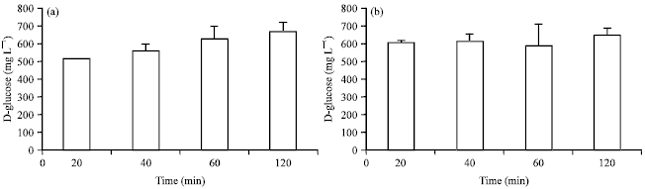
Technical Report
Application of the 2-Cyanoacetamide Method for SpectrophotometricAssay of Cellulase Enzyme Activity
Food Quality Laboratory, USDA-ARS, BARC-West, MD, Beltsville, United State of America
I. Vico
Institute of Phytomedicine, Faculty of Agriculture, University of Belgrade, Serbia
B.D. Whitaker
Food Quality Laboratory, USDA-ARS, BARC-West, MD, Beltsville, United State of America
V.L. Gaskins
Food Quality Laboratory, USDA-ARS, BARC-West, MD, Beltsville, United State of America
W.J. Janisiewicz
Appalachian Fruit Research Station, USDA-ARS, WV, Kearneysville, United State of America