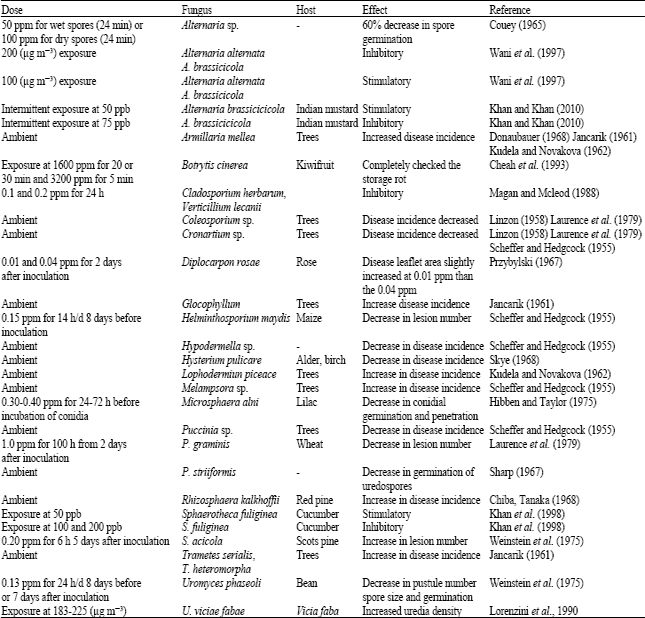
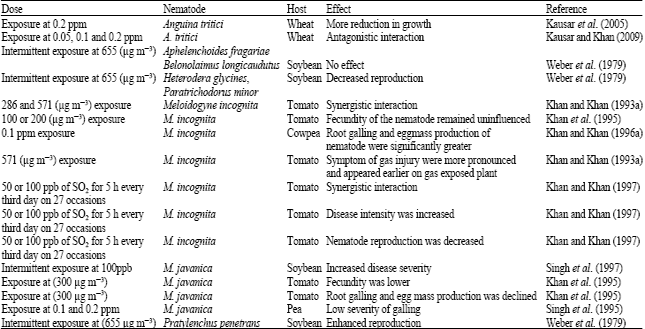
Review Article
Plants Response to Diseases in Sulphur Dioxide Stressed Environment
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh 202 002, India
Mohd Mahmud Khan
Department of Plant Protection, Faculty of Agricultural Sciences, Aligarh Muslim University, Aligarh 202 002, India