ABSTRACT
In this study, the effects of Arbuscular Mycorrhizal Fungi (AMF) Glomus mosseae (Gm), Glomus fasciculatum (Gf) and Rhizobium leguminosarum biovar phaseoli (Rlp), which are the important members of rhizosphere and biological control agents, were examined on the patho-system of Sclerotinia sclerotiorum (Lib.) de Bary (Ss) and common bean. The colonization and nodulation of two biological control agents exhibited differences as a result of reciprocal interactions of these items as well as the effect of the Ss. Nodulation of Rlp particularly decreased in triple inoculation. In addition, colonization of AMF significantly decreased in treatment of Ss+AMF than control AMF. Treatments of single inoculations of AMF and Rlp isolates reduced disease severity by 10.3-24.1%. It was determined that single biological control agents inoculations were more effective than dual inoculations (AMF+Rlp). When the morphological parameters of common bean were considered, all of the morphological parameters values were decreased in treatments which present pathogen isolate. Besides this, all biological control agents increased total contents of P and N in treated plants compared to the controls.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ppj.2009.74.78
URL: https://scialert.net/abstract/?doi=ppj.2009.74.78
INTRODUCTION
Arbuscular mycorrhizal fungi and rhizobia are two of the most important plant symbionts. They play a key role in natural ecosystems and influence plant productivity, plant nutrition and plant resistance (Demir and Akköpru, 2007). Mycorrhizas benefit the host through mobilization of phosphorus from nonlabile sources, whereas Rhizobium fixes N2 (Dar et al., 1997; Scheublin and Van der Heijden, 2006). The well-known activities of nitrogen-fixing bacteria improving the bioavailability of the major plant nutrients N and P, are very much enhanced in the rhizosphere of mycorrhizal plants where synergistic interactions of such microorganisms with mycorrhizal fungi have been demonstrated (Barea et al., 2002). A great deal of work has been carried out on the tripartite symbiosis legume-mycorrhiza-Rhizobium (Azcón-Aguilar and Barea, 1992; Barea et al., 2000, 2002). The inoculation of mycorrhizal fungi has been shown to improve nodulation and N2 fixation. Since soil-borne pathogens, as well as symbionts, share common habitat and show differential influence on the growth of the host plant, major interest has recently focused on the relevance of arbuscular mycorrhizas and rhizobia in the control of soil borne pathogens (Dar et al., 1997). As it known, these pathogens are typically difficult to control through conventional fungicide applications. By the arbuscular mycorrhizal fungi consistent reduction of disease symptoms has been described for fungal pathogens such as Phytophthora, Gaeumannomyces, Fusarium, Pythium, Rhizoctonia, Verticillium and Aphanomyces (Azcón-Aguilar and Barea, 1996; Demir and Akköpru, 2007). However, the effect of arbuscular mycorrhizas and Rhizobium on plant disease, nutrient uptake and rhizosphere microbial biomass and their activities are very difficult to generalize because the interactions involving arbuscular mycorrhiza, root rot fungi and Rhizobium vary with the microbial species and plant cultivars (Dar et al., 1997).
The present study was aimed to assess the roles of AMF and Rhizobium in the biological control of the common bean root and basal stem rot pathogen, Sclerotinia sclerotiorum, in addition to their influence on plant growth and soil nutrient availability.
MATERIALS AND METHODS
This study was conducted at Yuzuncu Yil University (Van/Turkey) in 2006 and 2007. Bean seeds (Phaseolus vulgaris cv. Sehirali beans) were surface sterilized in 2% sodium hypochlorite, rinsed 2-3 times in sterile distilled water and sown in plastic pots (16x18) containing a sterilized mixture of soil and sand (3:1, v/v). The following treatments, with five replications each, were included in the study: (1) G. mosseae (Gm); (2) G. fasciculatum (Gf); (3) Rhizobium leguminosarum biovar phaseoli (Rlp); (4); Sclerotinia sclerotiorum (Ss); (5) Gm+Rlp; (6) Gm+Ss; (7) Gm+Rlp + Ss; (8) Gf+Rlp; (9) Gf+Ss; (10) Gf+Rlp+Ss; (11) Rlp+Ss; (12) uninoculated control. All pots were placed in a growth chamber under standard conditions (14 h light (25-27°C), 65% relative humidity) in a completely randomized experimental design. Plants were watered twice a week with deionized water and 100 mL of the nutrient solution (containing 720 mg MgSO4.7H2O, 12.2 mg KH2PO4, 295 mg Ca (NO3)2.4H2O, 240 mg KNO3, 0.75 mg MnCl2.4H20, 0.75 mg KI, 0.75 mg ZnSO2.H2O, 1.5 mg H3BO3, 0.001 mg CuSO4.5H20, 4.3 mg FeNaEDTA and 0.00017 mg Na2MoO4.2H2O; modified from Vosátka and Gryndler (1999) was applied three times into each pot during the experiment (10 weeks).
Inoculations: Gm and Gf inoculations were accomplished by placing 2 g soil inoculum, containing 75 spores g-1 soil and 50 spores g-1 soil respectively, per pot, where the bean seeds were to be sown. Rhizobium, grown in yeast extract mannitol broth, was applied to seeds, containing 3.9x105 cells g-1. The seeds were sown immediately. Control plants did not receive any inoculum. For S. sclerotiorum inoculation, the wheat grains were boiled in tap water for 30 min. The boiled grains were put into 250 mL glass bottles up to half-length of the bottles and sterilized in an autoclave at 121°C for 40 min. A 5 mm disc agar covered with the mycelium of pathogen was placed in to the bottles containing the sterilized wheat grains. The bottles were incubated at 24°C for 4 weeks. After the pathogen has fully covered the grain surfaces with the mycelium the grains were then dried at room temperature. In addition, 15 sclerotia of pathogen were also added to the soil. Five grams of sterilized wheat grains were added to control pots (Irshad and Onogur, 2002).
Plant growth and nutrient uptake: At the end of the experiment, common bean plants were harvested 10 weeks after seed sowing. Plant roots were separated dried (70°C, 48 h) and weighed. Plants were analyzed for nitrogen and phosphorus by standard methods (Kacar, 1984) and nutrient uptake recorded.
Determination of disease severity caused by S. sclerotiorum, root colonization by AMF fungi and nodulation of Rhizobium: The disease development on each bean plant grown in the growth chamber for 10 weeks was rated by using the following scale (Irshad and Onogur, 2002): 0, no symptoms; 1, pathogen development on the basal stem, no symptoms in the plant tissue; 2, symptoms in the plant tissue, plant is still living; 3, plant died. Bean roots were dyed in order to determine the existence of Gm and Gf by a modified method of Phillips and Hayman (1970) and the colonization rates were determined by the Grid-Line Intersect Method (Giovanetti and Mosse, 1980). The number and activity of the nodules were recorded at the plant harvest (Ogut et al., 2003).
Statistical analysis: The data were subject to analysis of variance and means compared using Duncan’s multiple range tests.
RESULTS
Plant growth and nutrient uptake: Inoculation with arbuscular mycorrhizal fungi (Gm and Gf) and Rlp significantly increased shoot fresh weight and shoot dry weight, respectively, in comparison to the other treatments. Dry and fresh weights of root were significantly lower in control plants than those infected with Ss or inoculated with Gm, Gf, or Rlp either alone or in combination (Table 1). In the other hand, plant height was significantly increased when inoculated with Gm + Rlp.
Impacts of the biocontrol agents on the P and N contents and fresh and dry matter weights of tomato shoots and roots were also determined besides their effects on Ss. Mycorrhizal plants acquired more P, in comparison to the control, even in the presence of Ss (Table 1). The most remarkable results were obtained from the applications Gm+Rlp+Ss and Gm whose shoot and root contents were 72.5 and 80.8% higher, respectively than that of the control (Table 1). The N content of shoot in the single application of Rlp was higher than those in single or dual applications of the arbuscular mycorrhizal fungi (Table 1). The dual inoculation of Gm and Rlp had significant effect on the nitrogen content of roots (Table 1).
The number of nodules, AMF Root Colonization and Disease Severity: Colonization rates of the Gm and Gf and nodule number of Rlp are presented Table 2. As seen in Table 2, it was determined that Ss alone significantly (56.9, 53.2 and 57.1%, respectively) reduced the colonization levels of AM fungi and nodule number of Rlp compared with the non-treated control and the Ss treatments accompanied with the biological control agents (Gm+Rlp+Ss and Gf+Rlp+Ss) also reduced the colonization levels of arbuscular mycorrhizal fungi and nodule number of Rlp between 14.2-27.2%.
| Table 1: | Plant growth and nutrient uptake of the common bean inoculated with G. mosseae (Gm), G. fasciculatum (Gf), R. leguminosarum biovar phaseoli (Rlp) and S. sclerotiorum (Ss) alone or various combinations |
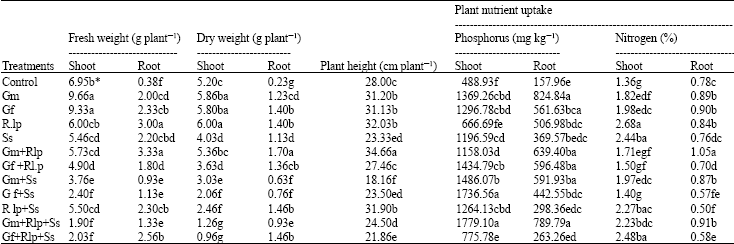 | |
| Values are means of five replications; Means within a column for each experiment by the same letter(s) are not significantly different (p<0.005) | |
| Table 2: | The number of nodules, AMF root colonization and disease severity of common bean plants inoculated with G. mosseae (Gm), G. fasciculatum (Gf), R. leguminosarum biovar phaseoli (Rlp) and S. sclerotiorum (Ss) alone or various combinations |
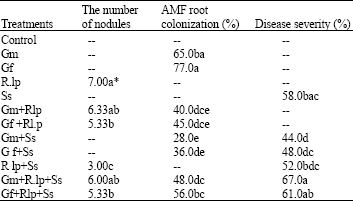 | |
| Values are means of five replications; Means within a column for each experiment by the same letter(s) are not significantly different (p<0.005) | |
Percentage efficacy of the mycorrhizal fungi and Rlp combinations with both single and dual inoculations against Ss are seen in Table 2. While the single inoculations of rhizosphere members inhibited Ss at the rate of between 10.3-24%, its dual inoculations increased disease severity rates of 4.9% and 13.4%.
DISCUSSION
Pathogenic infection by Ss reduced growth and nutrient uptake by the common bean, whereas inoculation with AMF or Rlp, alone, reduced pathogenicity. The decreased severity of disease and improved plant growth and nutrient uptake, expressed that the common bean inoculated by AMF or Rlp is somewhat resistant to the root pathogen Ss. Previous reports revealed that mycorrhizal plants offer increased tolerance to fungal root pathogens (Zambolin and Schenck, 1983; Jalali et al., 1983; Akköpru and Demir, 2005) and bacterization of legume seeds/seedlings with Rhizobium significantly reduced some rot root diseases caused by soil-borne fungal pathogens (Chakraborty and Purkayastha, 1984; Chakraborty and Chakraborty, 1989; Dar et al., 1997). It seems that enhanced plant growth improved nutrient assimilation and possibly a physical barrier have probably imparted altered resistance to the plants (Dar et al., 1997), as the disease incidence was reduced in plants inoculated with AM fungi and Rlp. Since the role of altered root exudates, changes in rhizosphere microbial activities and biochemical antagonism through phytoalexin and rhizobitoxine production as mechanisms of disease tolerance induced by AMF and Rlp (Dar et al., 1997) cannot conduct here.
AMF and Rhizobium, as the most important symbionts of rhizosphere have shown stimulating (Subba Rao et al., 1986; Champavat, 1990; Dar et al., 1997; Edwards et al., 1998) or inhibiting (Söderberg et al., 2002; Scheublin and Van der Heijden, 2006) effects on each other or on the growth of plants and pathogens. This was also confirmed in our study. In dual applications of both symbionts microorganisms decreased colonization or nodulation of each other. These inhibiting effects are thought to be related to the secretion of antimicrobial substances (Walley and Germida, 1997; Mar Vázquez et al., 2000). The colonization of AM fungi and the nodule numbers of Rlp was also significantly reduced presence of Ss. It is not clear how AMF root colonization or Rhizobium nodulation is affected by soil-borne pathogens (Johansson et al., 2004). However, it has been hypothesized that these effects may be related to the species and varieties of microorganisms and the conditions in the rhizosphere (Siddiqui and Shaukat, 2002; Anjair et al., 2003).
Legume being endowed with the unique ability to utilize the vast reservoir of atmospheric nitrogen with the help of symbiotic nitrogen fixing bacterium Rhizobium. Other soil microorganisms such as arbuscular mycorrhizal fungi have been credited for beneficial effects on plants. AMF absorbs phosphorus and translocates it to the roots (Champavat, 1990). Similar synergistic effect of Rlp and AM fungi has been recorded in common bean plants. It is also now well documented that inoculation of plants with AM fungi can stimulate nodulation and nitrogen fixation by legumes (Bethlenfalvay et al., 1982; Subba Rao et al., 1986; Champavat, 1990). Some workers showed that in P efficient soils, nitrogen fixation in several legumes inoculated with appropriate Rhizobium strain depended much on phosphorus, which could be supplied by mycorrhizal infection (Powell, 1976; Manjunath and Bagyaraj, 1984). The results reported also were confirmed in this study.
The present study concluded that suitable combinations of AM fungi and Rhizobium bacteria may increase the plant growth and resistance to pathogens. In future studies, therefore, more detailed investigations in various pathosystems and of the interactions between the microorganisms and the host plant are needed to develop much more efficient biocontrol of the related diseases.
ACKNOWLEDGMENTS
This study is a M.Sc. Thesis by Erizan Aysan under the supervision of Dr. Semra Demir. It was supported by Scientific Research Foundation of Yuzuncu Yýl University (2006-FBE-084).
REFERENCES
- Anjair, V., P. Cornelis and N. Koedom, 2003. Effect of genotype and root colonization in biological control of Fusarium wilts in pigeonpea and chickpea by Pseudomonas aeruginosa PNA1. Can. J. Microbiol., 49: 85-91.
Direct Link - Azcon-Aguilar, C. and J.M. Barea, 1997. Arbuscular mycorrhizas and biological control of soil-borne plant pathogens-an overview of the mechanisms involved. Mycorrhiza, 6: 457-464.
CrossRefDirect Link - Barea, J.M., R. Azcon and C. Azcon-Aguilar, 2002. Mycorrhizosphere interactions to improve plant fitness and soil quality. Antonie Van Leeuwenhoek, 81: 343-351.
Direct Link - Bethlenfalvay, G.J., R.S. Pacovsky, H.G. Bayne and A.E. Stafford, 1982. Interactions between nitrogen fixation, mycorrhizal colonization, and host-plant growth in the Phaseolus-Rhizobium-Glomus symbiosis. Plant Physiol., 70: 446-450.
PubMedDirect Link - Chakraborty, U. and R.P. Purkayastha, 1984. Role of rhizobitoxine in protecting soybean roots from Macrophomina phaseolina infection. Can. J. Microbiol., 30: 285-289.
CrossRefDirect Link - Chakraborty, U. and B.N. Chakraborty, 1989. Interaction of Rhizobium leguminosarum and Fusarium solani f.sp. pisi on pea-affecting disease development and phytoalexin production. Can. J. Bot., 67: 1698-1702.
CrossRefDirect Link - Dar, H., G., M.Y. Zargar and G.M. Beigh, 1997. Biocontrol of Fusarium root rot in the common bean (Phaseolus vulgaris L.) by using symbiotic Glomus mosseae and Rhizobium leguminosarum. Microb. Ecol., 34: 74-80.
PubMedDirect Link - Edwards, S.G., J.P.W. Young and A.H. Fitter, 1998. Interactions between Pseudomonas fluorescens biocontrol agents and Glomus mosseae, an arbuscular mycorrhizal fungus, within the rhizosphere. FEMS Microbiol. Lett., 166: 297-303.
Direct Link - Vazquez, M.M., S. Cesar, R. Azcon and J.M. Barea, 2000. Interactions between arbuscular mycorrhizal fungi and other microbial inoculants (Azospirillum, Pseudomonas, Trichoderma) and their effects on microbial population and enzyme activities in the rhizosphere of maize plants. Applied Soil. Ecol., 15: 261-272.
CrossRefDirect Link - Phillips, J.M. and D.S. Hayman, 1970. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc., 55: 158-161.
CrossRefDirect Link - Powell, C.L., 1976. Mycorrhizal fungi stimulate clover growth in New Zealand hill country soil. Nature, 264: 436-438.
CrossRefDirect Link - Scheublin, T.R. and M.G.A. van der Heijden, 2006. Arbuscular mycorrhizal fungi colonize nonfixing root nodules of several legume species. New Phytol., 172: 732-738.
CrossRefDirect Link - Siddiqui, I.A. and S.S. Shaukat, 2002. Resistance against the damping-off fungus Rhizoctonia solani systemically induced by the plant-growth-promoting Rhizobacteria Pseudomonas aeruginosa (IE-6S+) and P. fluorescens (CHA0). J. Phytopathol., 150: 500-506.
CrossRefDirect Link - Soderberg, K.H., P.A. Olsson and E. Baath, 2002. Structure and activity of the bacterial community in the rhizosphere of different plant species and the effect of arbuscular mycorrhizal colonisation. FEMS Microbiol. Ecol., 40: 223-231.
CrossRefDirect Link - Vosatka, V. and M. Gryndler, 1999. Treatment with culture fractions from Pseudomonas putida modifies the development of Glomus fistulosum mycorrhiza and the response of potato and maize plants to inoculation. Applied Soil Ecol., 11: 245-251.
CrossRefDirect Link - Walley, F.L. and J.J. Germida, 1997. Response of spring wheat (Triticum aestivum) to interactions between Pseudomonas species and Glomus clarum NT4. Biol. Fertil. Soils., 24: 365-371.
CrossRefDirect Link - Akkopru, A. and S. Demir, 2005. Biological control of Fusarium wilt in tomato caused by Fusarium oxysporum f.sp. lycopersici by AMF Glomus intraradices and some rhizobacteria. J. Phytopathol., 153: 544-550.
Direct Link









ForexTeacher Reply
Great job with the info. How did you find it? Please let me know.
MD.RAZIB HUMAUN Reply
It is a very nice paper which will help for further study.thanks to the auther.