Research Article
Characterization of Pepper yellow vein mali virus in Capsicum sp. in Burkina Faso
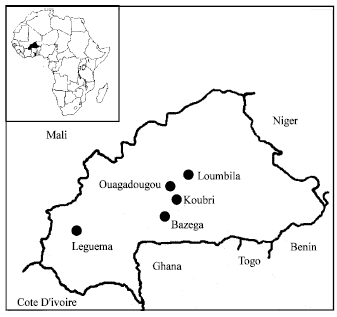
Laboratoire de Biochimie and Biologie Moleculaire, CRSBAN/UFR/SVT, Universite de Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso
V.S. Edgar Traore
Institut de l`Environnement et de Recherches Agricoles (INERA), 01 BP 476 Ouagadougou 01, Burkina Faso
Nicolas Barro
Laboratoire de Biochimie and Biologie Moleculaire, CRSBAN/UFR/SVT, Universite de Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso
Alfred S. Traore
Laboratoire de Biochimie and Biologie Moleculaire, CRSBAN/UFR/SVT, Universite de Ouagadougou, 03 BP 7021 Ouagadougou 03, Burkina Faso
Gnissa Konate
Institut de l`Environnement et de Recherches Agricoles (INERA), 01 BP 476 Ouagadougou 01, Burkina Faso
Oumar Traore
Institut de l`Environnement et de Recherches Agricoles (INERA), 01 BP 476 Ouagadougou 01, Burkina Faso