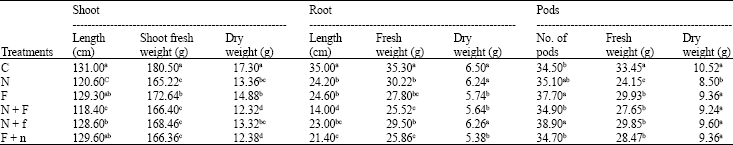
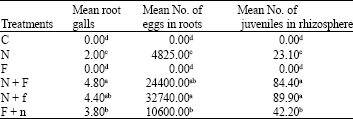
Research Article
Separate and Combined Effects of Fusarium oxysporum f. sp. tracheiphilum and Meloidogyne incognita on Growth and Yield of Cowpea (Vigna unguiculata L. Walp) Var. Moussa-Local
Unit of Plant Science and Biotechnology, Department of Biological Sciences, Nasarawa State University, P.M.B. 1022, Keffi, Nigeria