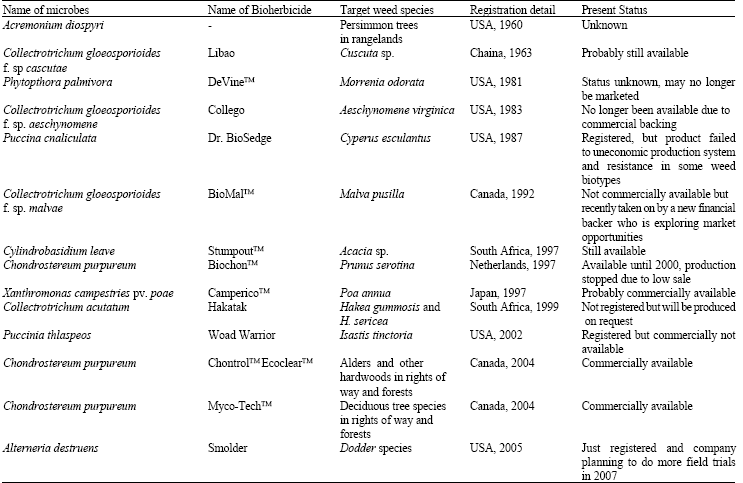
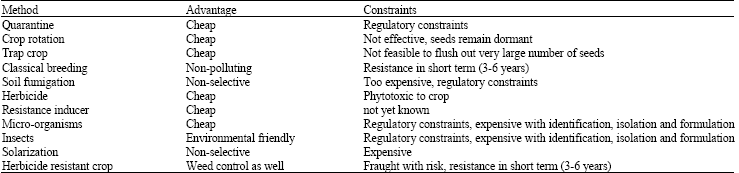
Research Article
Microbial Herbicides for Weed Management: Prospects, Progress and Constraints
MAEP Division, North East Institute of Science and Technology, Formerly Regional Research Laboratory (CSIR), Jorhat-785006, India
J.J. Mahanta
MAEP Division, North East Institute of Science and Technology, Formerly Regional Research Laboratory (CSIR), Jorhat-785006, India
N. Bhattacharyya
Department of Biotechnology, Gauhati University, Assam-781 014, India
M. Bhuyan
MAEP Division, North East Institute of Science and Technology, Formerly Regional Research Laboratory (CSIR), Jorhat-785006, India
P. Boruah
MAEP Division, North East Institute of Science and Technology, Formerly Regional Research Laboratory (CSIR), Jorhat-785006, India
T.C. Sarma
MAEP Division, North East Institute of Science and Technology, Formerly Regional Research Laboratory (CSIR), Jorhat-785006, India