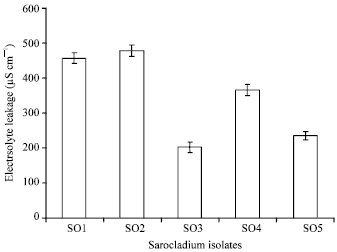
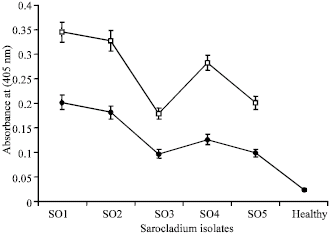
Research Article
Variation in Toxin Production among Isolates of Sarocladium oryzae, the Rice Sheath Rot Pathogen
Department of Plant Pathology and Crop Physiology, Louisiana State University Agricultural Center, Baton Rouge, LA 70803, USA
S. Babu
Department of Microbiology, Immunology and Biochemistry, North Eastern Ohio Universities, College of Medicine, Rootstown, OH44272, USA
G. Amutha
Department of Plant Pathology, Centre for Plant Protection Studies, Tamil Nadu Agricultural University, Coimbatore 641003, India
T. Raguchander
Department of Plant Pathology, Centre for Plant Protection Studies, Tamil Nadu Agricultural University, Coimbatore 641003, India
R. Samiyappan
Department of Plant Pathology, Centre for Plant Protection Studies, Tamil Nadu Agricultural University, Coimbatore 641003, India