Research Article
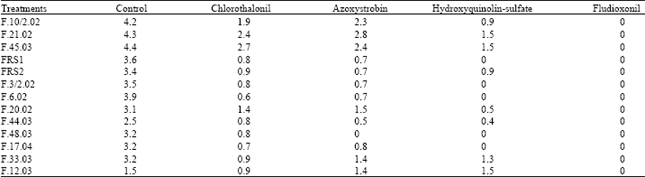
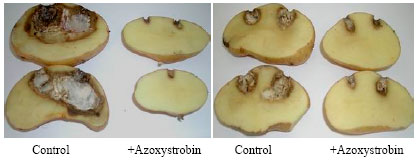
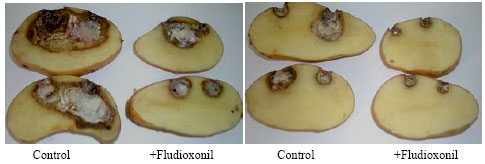
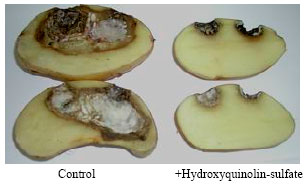
In vitro and in vivo Interaction of Four Fungicides with the Fusarium Species Complex Causing Tuber Dry Rot in Tunisia
National Institute of Agronomic Research of Tunisia, PRRDA-CE 4042 Chott-Mariem, 4042, Sousse, Tunisia
H. Jabnoun-Khiareddine
High School of Horticulture and Breeding of Chott-Mariem, 4042, Sousse, Tunisia
F. Ayed
High School of Horticulture and Breeding of Chott-Mariem, 4042, Sousse, Tunisia
M. El Mahjoub
High School of Horticulture and Breeding of Chott-Mariem, 4042, Sousse, Tunisia