ABSTRACT
Nine Streptomyces strains were screened for their potential to protect Cucumis sativus from Cucumber Mosaic Virus (CMV). Foliage treatment with the Streptomyces culture filtrates resulted in 50-85% reduction of the mosaic symptoms. S. violatus, S. nasri H35, Streptomyces sp., S. aureofaciens and S. violaceuisniger showed the highest five antiphytoviral activities. The filtrate of the five Streptomyces strains showed more antiphytoviral activity when applied before virus inoculation compared to their application after viral inoculation. The most favourable incubation period for the highest antiphytoviral activity detected in the filtrates were attained after incubation for 6 h. On the other hand, soaking of seeds for 2 h in the actinomycetes filtrates resulted in the highest viral inhibition, although there is no direct relationship between the percentage of inhibition and the time of soaking. Generally, S. violaceusniger filtrate recorded the highest percentage of viral inhibition (85%). Studies on the ultrastructural changes in cucumber plants showed that viral inoculation led to distortion of spongy and vascular bundle cells as a results of presence of large vacuoles inside these cell. Treatment of inoculated cucumber plants with either Streptomyces strains gave a moderate recovery of cells.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ppj.2006.343.349
URL: https://scialert.net/abstract/?doi=ppj.2006.343.349
INTRODUCTION
Viral diseases are responsible for the major concern among agriculturists because of their economic impact on crop production (Hansen, 1989; Mink, 1993). Viruses were responsible for some unusual effects on crops such as their need for greater nutrient requirements as in fruit trees and reduced coloration in grapes which adversely affect the fresh market sales (Fraizier and Mellor, 1970; Goheen, 1970; Thomas, 1976; Duffus, 1977; Vander Zaag, 1987; Kozeletskaia et al., 2003). Several plant diseases were reported to be caused by viruses. Among these were barely yellow dwarf virus (Gill, 1980), tomato spots (Cho et al., 1989), potato leaf roll (Jellis and Boulton, 1984), cassava mosaic virus (Thomas, 1976) and rice tungro virus (Hasanuddin and Hibino, 1989).
Viruses are also responsible for a wide range of economic losses to the most human food and economic crops world wide, especially in Egypt. Cucurbit crops are subject to severe losses due to several potyviruses (Grumet et al., 2000), including the highly aggressive Cucumber Mosaic Virus (CMV). Different viral isolates were found in infected cucurbits throughout the world including localities in Europe, America, Australia and Asia (Desbiez and Lecoq, 1997; Prieto et al., 2001).
Streptomyces strains were the source of many useful and consequently profitable antibiotics (Williams et al., 1989; Lazzarini et al., 2000; El-Naggar et al., 2003) and antiviral agents (Yeo-woonHyung et al., 1997; Habib et al., 2001; Ghaly et al., 2005; Sonya et al., 2005). The great majority of preparations that showed antiviral activity had been isolated from streptomycetes. Among the antiviral agents isolated from actinomycetes, cytovirin, cephalomycin, heliomycin, myxoviromycin, niromycin, olivomycin and quinomycin. In tobacco (Nicotiana tabacum CV xanthin), actinomycin D reduced the resistance to TMV that was induced by ployacrylic acid or by earlier infection with PVY (Kassanis and White, 1974). Several antiviral agents were isolated from streptomycetes broth, the broth of S. rochei could inhibit TMV (from leaves of Datura metel), while the broth of S. rimosus or S. gougerotti caused a weak inhibition (Mansour et al., 1988). Galal and El-Shirbiny (1995) demonstrated the effect of different concentrations of caeseorhodomycin (produced by S. caeseorhodomyces) on the inhibition of potato virus X of D. stramonium plant and the rate of inhibition was concentration dependent. Sacrmento et al. (2004) isolated strain 606 identified as Streptomyces sp. and found to possess detectable antiviral activity capable of inhibiting the propagation of an acyclovir resistant herpes simplex virus type 1 strain on HEP-2 cells at non cytotoxic concentration.
The Systemic Acquired Induced Resistance (SAR) by biotic or a biotic agents had been recognized to play an important role in defense against plant viruses, since this resistance was mainly associated with the introduction of novel proteins (Ponz and Bruening, 1986; Faccioli et al., 1994) in treated plants which was the actual virus inhibitory proteins. These proteins thus induced antiviral state in plants through formation of de novo synthesized protein and perhaps were active in signaling the activation of defense mechanism in susceptible hosts and hence had been called systemic resistance inducers and the novel proteins induced resembled ribosome-inactivating proteins (Verma and Varsha, 1995).
The present study is aimed to screen 9 streptomycete isolates for their natural antiphytoviral activities against Cucumber Mosaic Virus (CMV) which produce a systemic viral infection on Cucumis sativus plants.
MATERIALS AND METHODS
This study was conducted in green house of Biology Department, Faculty of Science, Taibah University, Almadinah Almunawwarah, Kingdom of Saudi Arabian in the year of 2005.
Seeds of cucumber (Cucumis sativus L.) cv. beit alpha were kindly provided by the seed bank, Agriculture Research Institute (ARI), Dokki, Giza, Egypt. Naturally infected leaves of cucumber showing mosaic and yellowing symptoms were collected from fields in Al-Madinha Al-Munawarrah, Kingdom of Saudi Arabia. A fertile loamy soil was used for cultivation of cucmber plants.
Streptomyces strains and growth conditions: Streptomyces isolates were S. nasri YG62, S. nasri mutants H5, H6, H15, H35, S. orientalis, S. cratifers, S. aureofaciens and S. violaceusniger. Starch nitrate medium was used for the growth of Streptomyces strains at 30°C for 10 days for the detection of the antiviral activity. Starch-nitrate agar medium was used for the maintenance of Streptomyces strains (Hassan et al., 2001). The pH was adjusted at 7.2.
Antiphytoviral activities
Cultivation of cucumis sativus: Seeds of host plants (Cucumis sativus) were cultivated in plastic pots (30 cm) each containing 2 kg fertile loamy soil; autoclaved at 1.5 atm pressure for 120 min before cultivation, at 1 cm depth below the soil surface and kept under the natural daylight. Irrigation of cultivated seeds was carried out regularly until the end of each experiment.
Preparation of virus inoculum: Five grams of naturally infected cucumber plantlets with cucumber mosaic virus symptoms (CMV) were ground in sterile mortar and pestle and homogenized with 5 mL of 0.01 M phosphate buffer, pH 7.2, then filtered by Whatman filter paper No. 1. The volume was made up to 20 mL with phosphate buffer (pH 7.2) and kept at -20°C until use according to the method described by Lisa et al. (1981). Pathogenicity test was done on healthy cucumber plants. Inoculation was done after dusting cotyledonary leaves with 600 meshes carborandum. After interval of 3 weeks, the external symptoms of virus infection similar to those found in the naturally infected leaves were noticed.
In vitro effect of culture filtrates on the infectivity of CMV: Two milliliter aliquots of Streptomyces filtrate were mixed with viral sap for 10 min (1:1 v/v) and then applied directly onto cotyledonary cucumber leaves. Two control tests were done. The first: leaves of cucumber were inoculated with CMV. The second: control leaves were inoculated with the buffer only. The symptoms were observed and recorded after 3 weeks. The mean percentage of viral control and the percentage of inhibition were determined. Plants were harvested after 3 weeks.
In vivo effect of culture filtrate application on the infectivity of CMV
Post-inoculation treatment of leaves: The cotyledonary leaves of cucumber were inoculated with CMV after dusting the leaves with carborandum (600 meshes) and then the inoculated leaves washed with sterile distilled water. The Streptomyces filtrates were applied on the inoculated plants after 1, 6, 12, 24, 36, 48 and 72 h. Control plants were inoculated with CMV or treated with phosphate buffer alone. The symptoms were observed and recorded after 3 weeks post infection.
Pre-inoculation treatment of leaves: The Streptomyces filtrates of 9 isolate were applied before the inoculation of cucumber plants with CMV at 1, 6, 12, 24, 36, 48 and 72 h. Control plants include virus-infected plants and phosphate buffer treated plants. The symptoms were observed after 3 weeks of virus inoculation. The mean percentages of inhibition were calculated.
Seed soaking of Cucumis sativus with Streptomyces filtrates before cultivation: Sixty seeds of Cucumis sativus were soaked in 100 mL of each Streptomyces filtrates for different periods of 2, 4 and 6 h. For each soaking period, 20 seeds were cultivated. Control seeds were soaked in distilled water for similar time intervals. After germination, the cotyledonary leaves were rubbed with CMV by mechanical inoculation and the inoculated leaves were washed with distilled water. The symptoms were observed and recorded after 3 weeks. The percentages of inhibition were calculated.
Electron microscopy: CMV-infected, filtrate-treated and healthy leaves were processed for transmission electron microscopy (TEM) according to Perera and Gay (1976). The cucumber leaves were cut into pieces (1 mm square) and fixed in 2.5% (w/v) glutaraldehyde in 0.1 M sodium cacodylate (pH 7) at room temperature. After 24 h, the specimens were washed three times with glutaraldehyde and fixed in 1% (v/v) osmium tetraoxide/sodium cadodylate buffer for 1 h. Tissues washed 2-3 times and then dehydrated in alcohol (45, 60 and 75%) for 10 min each. Specimens were then soaked in epoxy propane-ethanol solution (1:1 v/v) and left to stand for 30 min. In closed container, specimens were transferred into pure epoxy propane and spurs resin (1:1 v/v) and left for 1 h. The lids of the cups were left slightly open, allowing epoxy propane to evaporate over night. After 3 days, tissue fragments were embedded, in fresh resin and left in an oven at 60°C for 48 h to polymerize.
Ultra-thin sections were made using Reichter ultra microtome. Thin sections were picked up on a dull surface of formvar (polyvinyl/formaldehyde)-coated copper grids. Ultra-thin sections were stained using 2% uranyl acetate (Juniper et al., 1970) for 30 min and then washed gently and dried. The sections were examined and photographed using Jeol 100 electron microscope.
RESULTS
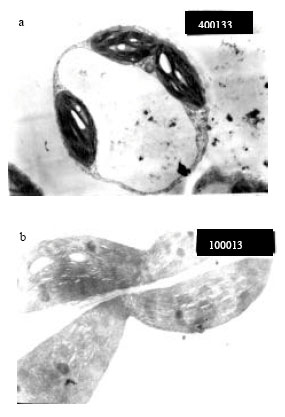
Screening for the antiphytoviral activities: Nine Streptomyces strains were screened for their antiphytoviral activity. The filtrates of these strains were mixed with an equal volume (v/v) of virus containing sap (CMV) and inoculated on cultivated Cucumis sativus preliminary leaves. The viral symptoms appeared on seedlings aged 3 weeks compared with their respective controls. The infected plant leaves are shown in Fig. 1b as compared with healthy plant leaves (Fig. 1a) and the transmission electron microscopy revealed chloroplast changes centered around vacuolation as shown in Fig. 2. All filtrates had inhibitory effect but with different percentages (Table 1). Five Streptomyces strains (S. violatus, S. nasri H35, S. sp., S. aureofaciens, S. violaceusniger) had the highest antiphytoviral activities (70-85%) while S. nasri H6 and S. orientalis had the lowest antiphytoviral activities.
 | |
| Fig. 1: | Healthy cucumber plant (a) and plants infected with CMV (b) showing yellow mosaic symptoms |
 | |
| Fig. 2: | Electron micrographs of ultra thin section of (a) CMV- infected leaf of Cucumin sativus showing vacuolation and (b) healthy leaf of Cucumis sativus showing a normal chloroplast |
| Table 1: | In vitro effect of different culture filtrates (filtrate-viral mixture) of streptomycete isolates on the percentage of CMV inhibition |
 | |
| Viral inhibition% = a-b/a x 100, where a represents the number of inoculated plants (20 plants) and b represents the number of plants showing viral symptoms | |
In vivo effect of culture filtrates before and after virus inoculation
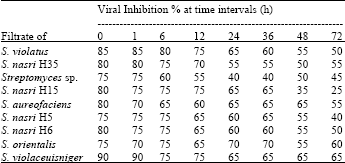
Pre-inoculation treatment: The effect of the most active 5 filtrates on the infectivity of CMV before inoculation on Cucumis sativus leaves for different periods was investigated (Table 2). Each filtrate was applied on the cotyledonary leaves of Cucumis sativus plant which was inoculated with CMV at different time intervals (1, 6, 12, 24, 36, 48 and 72 h). The plants were left in the green house for 3 weeks till the symptoms were developed. The percentage of inhibition was calculated. The filtrates produced a remarkable inhibition at 6 h of filtrate application. The inhibitory effect was gradually decreased at 12-72 h before virus inoculation. S. violaceusniger filtrate was the best viral inhibitor followed by S. violatus, S. nasri H6, S. nasri H15, S. nasri H35 and S. aureofaciens.
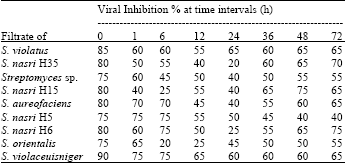
Post-inoculation treatment: Data show the effect of application of each of the 5 culture filtrates on the two primary leaves of Cucumis sativus plant after inoculation with CMV at different intervals from 1-72 h are present in (Table 3). S. violaceusniger filtrate was the best viral inhibitor followed by S. violatus, S. nasri H6, S. nasri H15, S. nasri H35 and S. aureofaciens. The foliar application of actinomycete filtrates before viral infection were generally higher than after viral inoculation.
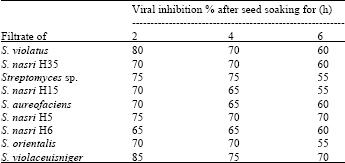
Infectivity pattern after seeds soaking in Streptomyces culture filtrates: Results in Table 4 showed the effect of soaking of seeds for 2, 4, 6 h with the streptomycetes filtrates. Variation was noticed in viral inhibition, but there is no relation between percentage of inhibition and the time of soaking. The shortest time of soaking showed higher viral inhibition. S. violaceusniger recorded the highest percentage of viral inhibition for seeds soaked in tested filtrates for 2 and 4 h; giving inhibition of 85, 75 and 70%, respectively.
| Table 2: | In vivo effect of pre-inoculation of cucumber leaves with streptomycete filtrates on the infectivity of CMV at different time intervals |
 | |
| Table 3: | In vivo effect of post-inoculation of cucumber leaves with streptomycete filtrates on infectivity of CMV at different time intervals |
 | |
| Table 4: | Effect of soaking tested seed in streptomycetes filtrates on the infectivity of with CMV on cucumber plants |
 | |
| 20 seeds were used for each treatment, Viral Inhibition% = a-b/a x 100, where a represents the number of control seeds soaked in distilled water, b = No. of infected plants from seeds soaked in streptomycete filtrates | |
DISCUSSION
Great economic losses occur worldwide due to the viral plant diseases. Losses are often more insidious, frequently less conspicuous and therefore go unnoticed and untreated (Waterworth and Hadidi, 1998). Cucumber Mosaic Virus (CMV) is one of the most economically important viruses of cucurbit crops, which could have contributed to its rapid spread worldwide. Soil is a rich source of streptomycetes for being able to utilize a wide variety of organic compounds. They are known to produce many secondary metabolites useful in agricultural, medicinal and biochemistry fields.
The present data showed that all the tested isolates had a variable inhibitory effect on CMV infected cucumber plants. Mansour et al. (1988) could isolate antiviral agents from S. rimosus and S. gougerotti against TMV. Also Galal and El-Shirbiny (1995) reported that caeseorhodomycin produced by S. caeseorhomyces had an antiviral activity against potato virus X on Datura stramonium plants. Yassin and Galal (1998) reported that the filtrate of some fungi and actinomycetes had an antiviral activities against tobacco necrosis virus.
The data showed that application of the streptomycetes filtrates on cucumber leaves at different periods before infection with CMV led to an inhibitory effect, the highest effect was obtained after 6 h of application. The variation in viral inhibition percentages may reflect the variation in resistance against CMV which may be due to the presence of different metabolites in the microbial filtrates which may have an inductive effect in the host plant to inhibit the infection of CMV (Kegler et al., 1993; Galal and El-Shirbiny, 1995; Ghaly et al., 2005). Similar results were reported by Mansour et al. (1988) and Shoman et al. (2003). These inducers could be proteins in nature as those produced in Borhaavia diffusa (Verma and Awasthi, 1980) and Clerodendium aculeatum (Verma and Varsha, 1995) and inhibited the virus infection. Barakat (2001) indicated that the time course was essential to accumulate such proteins and the induction of systemic resistance is non specific response effective against a broad spectrum of viruses.
Gaborjanyi and Tobias (1986) suggested that the inhibitor may remain active on leaf surface, enter the leaf together with the virus and cause their inhibitory activity inside the leaf, or it may alter cell metabolism to affect late virus replication (Fisher and Nienhaus, 1973; Wilson and Miles, 1975; Loebenstein and Gera, 1981; Nogajan et al., 1997; Balasarawathi et al., 1998). Similar results were reported by several workers (Pandey and Mohan, 1986; Patel and Reynolds, 1992) where Spinacia extracts showed considerable inhibition when applied for 1 to 3 h after virus inoculation. The action of antiviral substances was host dependent and acted on the host plant rather than the virus (Fischer and Nienhaus, 1973; Verma et al., 1979). However, the percentage of inhibition in the present data was not significantly reduced especially when the filtrates were applied after 6 h and up to 72 h after CMV inoculation. This may be due to the great opportunity for virus penetration and replication through the host cells.
Similar results and discussion were obtained by Van Wees et al., 2000 which showed that the combined induction of Induced Systemic Resistance (ISR) and Systemic Acquired Resistance (SAR) by specific strain of growth promoting rhizobacteria afforded greater protection of Arabidopsis plants against broad spectrum of pathogens than each alone, indicating that ISR and SAR can act in enhancing resistance to pathogens. Systemic Acquired Resistance (SAR) had been previously shown in many infected plants by several authors (Parent and Asselin, 1984; Ye et al., 1990). Other workers reported that SAR could be induced by plant extracts (Verma and Awasthi, 1980; Verma et al., 1979; Neeta and Verma 1995; Dhirendra et al., 1997).
Treatment with tested filtrates containing crude antiviral substance pre-inoculation with CMV were found to be effective than post-inoculation. Similar observations were found by many other workers; Verma et al. (1979) found that inhibition of TMV infection was highly significant when Datura metal leaf extract was applied 24 h before virus inoculation. These results were in agreement with Rao and Shukla (1985) who reported that aqueous extracts of dry Coconut showed significant antiviral activity against PVY when applied 24 h before virus inoculation and no inhibition was observed when extract was applied 24 h after virus inoculation.
The present data showed that soaking of Cuumis sativus seeds in the filtrate of the Streptomyces strains for 2, 4 and 6 h reduced CMV infectivity. The highest inhibition of CMV was achieved after 2 h. Hundreds of viruses infect seeds and many are transmitted to the resulting seedlings (Mink, 1993). The infected seed may appear normal depending on virus and host species, but sometimes seeds looked discolored, cracked or reduced in size, weight, or viability. Maurhoffer et al. (1994) suggested that the seed soaking treatment was an important to protect plants against foliar pathogens.
In conclusion, the pre-inoculation of cucumber plant with the filtrates had the higher antiphytoviral effect against CMV followed by soaking the Cucumis sativus seeds with the tested filtrates for 2 h. S. violaceusniger proved to be the ideal strain to control this viral infection in this study. The compound(s) produced by this strain and the mode of action are currently undertaken and will be reported later.
REFERENCES
- Balasarawathi, R., S. Sadasiram, M. Ward and J.M. Walker, 1998. An antiviral protein from Bauganivella spectabilis roots. Purification and characterization. Phytochemistry, 74: 1561-1565.
CrossRefDirect Link - Desbiez, C. and H. Lecoq, 1997. Zucchini yellow mosaic virus. Plant Pathol., 46: 809-829.
Direct Link - El-Naggar, M.Y., M.A. Hassan, W.Y. Said and S.A. El-Assar, 2003. Effect of support materials on antibiotic MSW2000 production by immobilized Streptomyces violatus. J. Gen. Applied Microbiol., 49: 235-243.
PubMedDirect Link - Ghaly, M.F., N.M. Awny, A.M. Galal and A. Askora, 2005. Characterization and action of antiphytoviral agent produced by certain streptomycetes species against zucchini yellow mosaic virus. Egypt J. Biotechnol., 19: 209-223.
Direct Link - Grumet, R., Z. Ullah, A. Gal-on and R. Raccah, 2000. Cucurbit-potyviurs interactions influencing virus movement and accumulation. Acta Hortic., 510: 327-334.
Direct Link - Habib, E.S.E., K. Yokomizo, K. Nagao, S. Harada and M. Uyeda, 2001. Antiviral activity of fattiviracin Fr-8 against human immunodeficiency virus types 1(HIV-1). Biosci. Biotechnol. Biochem., 65: 683-685.
Direct Link - Hansen, A.J., 1989. Antiviral chemicals for plant disease control. Crit. Rev. Plant Sci., 8: 45-88.
Direct Link - Hassan, M.A., M.Y. El-Naggar and W.Y. Said, 2001. Physiological factors affecting the production of an antimicrobial substance by Streptomyces violatus in batch culture. Proceedings of the 1st International Conference of Egyptian British Biological Society: Biodiv. Conserv. Natural Heritage, Jul. 29-30, Ismailia, Egypt, pp: 1-10.
- Lazzarini, A., L. Cavaletti, G. Toppo and F. Marinelli, 2000. Rare genera of Actinomycetes as potential producers of new antibiotics. Antonie van Leeuwenhoek, 78: 399-405.
PubMed - Lisa, V., G. Boccardo, G. D'Agostino, G. Dellavalle and M. d'Aquilio, 1981. Characterization of potyvirus that causes zucchini yellow mosaic. Phytopathology, 71: 667-672.
Direct Link - Loebenstein, G. and A. Gera, 1981. Inhibitor of virus replication released from tobacco mosaic virus-infected protoplasts of a local lesion-responding tobacco cultivar. Virology, 114: 132-139.
PubMed - Maurhoffer, M., C. Hase, J.P. Matraux and G. Defago, 1994. Induction of systemic resistance of tobacco to tobacco necrosis virus by the root-colonizing Pseudomonas fluorescens strains CHAO: Influence of the gacA gene and pyoverdine production. Phytopathology, 84: 139-146.
Direct Link - Mink, G.I., 1993. Pollen and seed transmitted viruses and viroids. Ann. Rev. Phytopathol., 31: 375-402.
PubMedDirect Link - Neeta, S. and H.N. Verma, 1995. Chenopodium mural leaf extract, a potent virus inhibitor. Ind. Phyathol., 48: 177-179.
Direct Link - Nogajan, N., H.R. Reddy and K.S. Ravi, 1997. Effect of exogenously applied plant products on peper vein banding virus transmission, multiplication and symptom production in bell peper (Capsicum annum L.) Ind. J. Virol., 13: 161-163.
Direct Link - Parent, J.G. and A. Asselin, 1984. Detection of pathogenesis related proteins (PR or b) and of other proteins in the intercellular fluid of hypersensitive plants infected with tobacco mosaic virus. Can. J. Bot., 62: 564-569.
Direct Link - Ponz, F. and G. Bruening, 1986. Mechanism of resistance to plant viruses. Ann. Rev. Phytopathol., 24: 355-381.
CrossRefDirect Link - Prieto, H., A. Brunan, P. Hinirichsen and C. Munoz, 2001. Isolation and molecular characterization of a chilean isolate of zucchini yellow mosaic virus. Plants Dis., 85: 644-648.
Direct Link - Sacramento, D.R., R.R.C. Rosalie, D.W. Marcia, F.T. Luiz and G. Marta et al., 2004. Antimicrobial and antiviral activities of an actinomycete (Streptomyces sp.) isolated from a Brazilian tropical forest soil. World J. Microbiol. Biotechnol., 20: 225-229.
Direct Link - Shoman, S.A., N.A. Abd-Allah and A.F. El-Baz, 2003. Induction of resistance to tobacco necrosis virus in bean plants by certain microbial isolates. Egypt. J. Bot., 5: 10-18.
Direct Link - Sonya, H.M. and A.M. Galal, 2005. Identification and antiviral activities of some halotolerant streptomycetes isolated from Qaroon Lake. Int. J. Agric. Biol., 7: 747-753.
Direct Link - Verma, H.N. and L.P. Awasthi, 1980. Occurrence of a highly antiviral agent in plants treated with Boerhaavia diffusa inhibited. Can. J. Bot., 58: 2141-2144.
Direct Link - Ye, X.S., S.Q. Pan and J. Kue, 1990. Activity, isozyme pattern and cellular localization of peroxidase or related to systemic resistance of tobacco to blue mold (Peronospora tabacino) and to tobacco mosaic virus. Phytopathology, 80: 1295-1299.
Direct Link - Yeo Woon, H., K. Youngho, P. Eunkyungi, K. Sangseock and W.H. Yeo et al., 1997. Physicochemical characteristics and antiviral activity of ASA, an antibiotic produced by actinomycetes B 25. Korean J. Plant Pathol., 13: 63-68.
Direct Link








