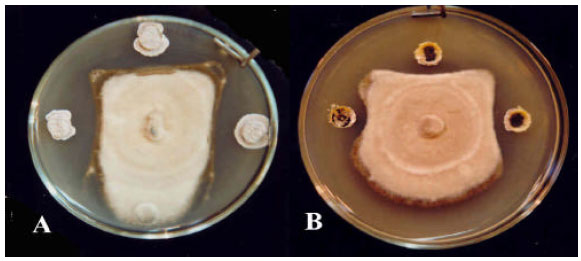
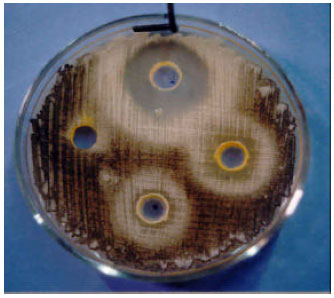
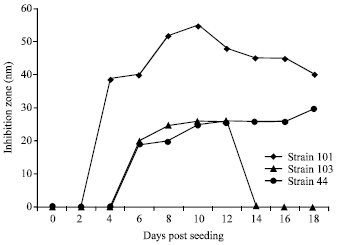
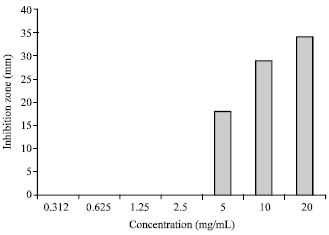
Research Article
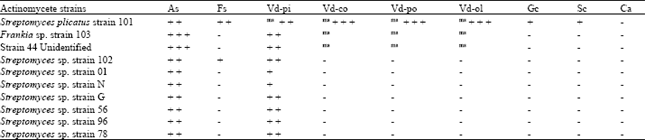
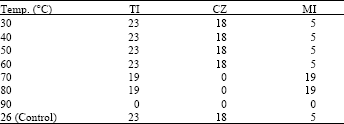
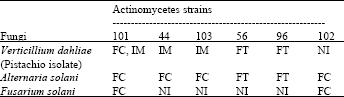
Antifungal Characterization of Actinomycetes Isolated from Kerman, Iran and Their Future Prospects in Biological Control Strategies in Greenhouse and Field Conditions
Department of Plant Pathology, College of Agricultural Sciences, Bahonar University of Kerman, Iran
Parvin Rashid Farrokhi
Department of Agronomy and Plant Breeding, College of Agricultural Sciences, Bahonar University of Kerman, Iran
Sonia Aghighi
Department of Plant Pathology, College of Agricultural Sciences, Bahonar University of Kerman, Iran
Leyla Shahidi Bonjar
Department of Plant Pathology, College of Agricultural Sciences, Bahonar University of Kerman, Iran
Ataollah Aghelizadeh
Management and Planning Organization of Kerman Province, Kerman, Iran