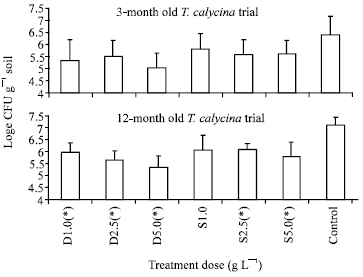
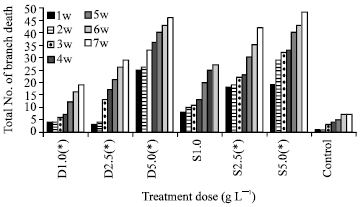
Research Article
Phosphonate (PO3-) Effectiveness Against Phytophthora cinnamomi Rands on Thryptomene calycina, Banksia grandis and Banksia spinulosa
Department of Biology and Research Development Group on Life Sciences, Institut Teknologi Bandung, Jalan Ganesha 10, Bandung 40132, Indonesia
David I. Guest
School of Botany, The University of Melbourne, Victoria-Australia