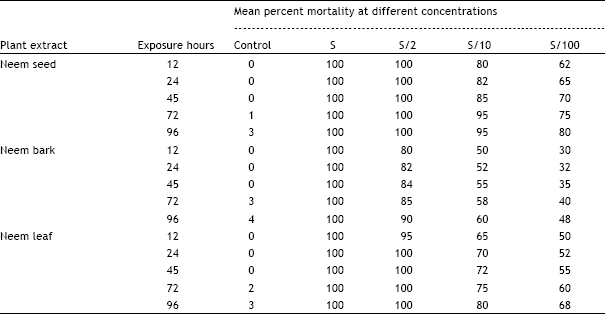
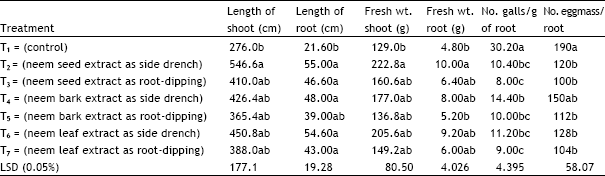
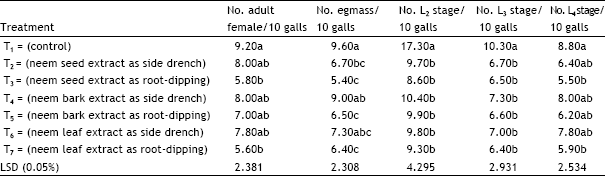
Research Article
Use of Neem Extract in Controlling Root-knot Nematode (Meloidogyne javanica) of Sweet-gourd
Horticulture Research Centre, Bangladesh Agricultural Research Institute, Gazipur, Bangladesh,
M.H. Rashid
Horticulture Research Centre, Bangladesh Agricultural Research Institute, Gazipur, Bangladesh,
M. Nazim Uddin
Horticulture Research Centre, Bangladesh Agricultural Research Institute, Gazipur, Bangladesh,
M.S. Hossain
Horticulture Research Centre, Bangladesh Agricultural Research Institute, Gazipur, Bangladesh,
M.E. Hossain
World Vision, Phulpur, Mymensingh, Bangladesh
M.U. Ahmed
Department of Plant Pathology, Bangladesh Agricultural University, Bangladesh