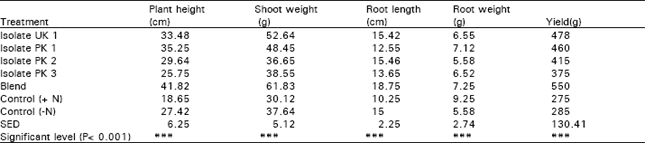
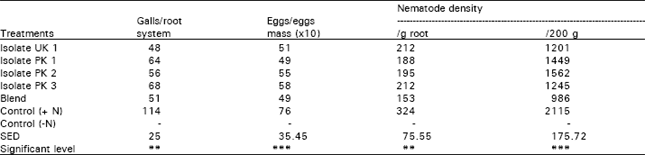
Research Article
Field Evaluation of Pasteuria Isolates for the Control of Root-Knot Nematodes, Meloidogyne javanica on Tomato
Biocontrol Research Laboratory, Department of Botany, University of Karachi, Karachi-75270, Pakistan Department of Plant Pathology, University of Agriculture, Faisalabad, Pakistan
Nazir Javed
Biocontrol Research Laboratory, Department of Botany, University of Karachi, Karachi-75270, Pakistan Department of Plant Pathology, University of Agriculture, Faisalabad, Pakistan
M. Javed Zaki
Biocontrol Research Laboratory, Department of Botany, University of Karachi, Karachi-75270, Pakistan Department of Plant Pathology, University of Agriculture, Faisalabad, Pakistan