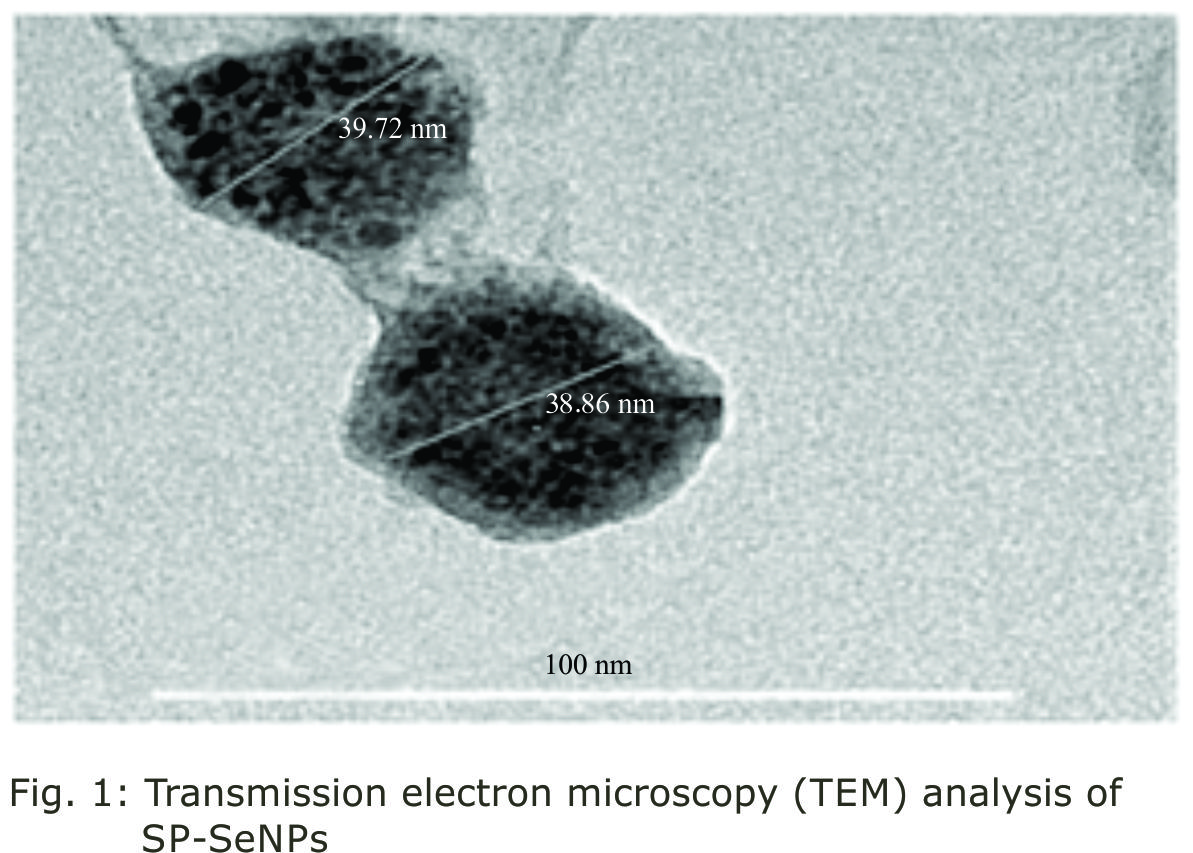
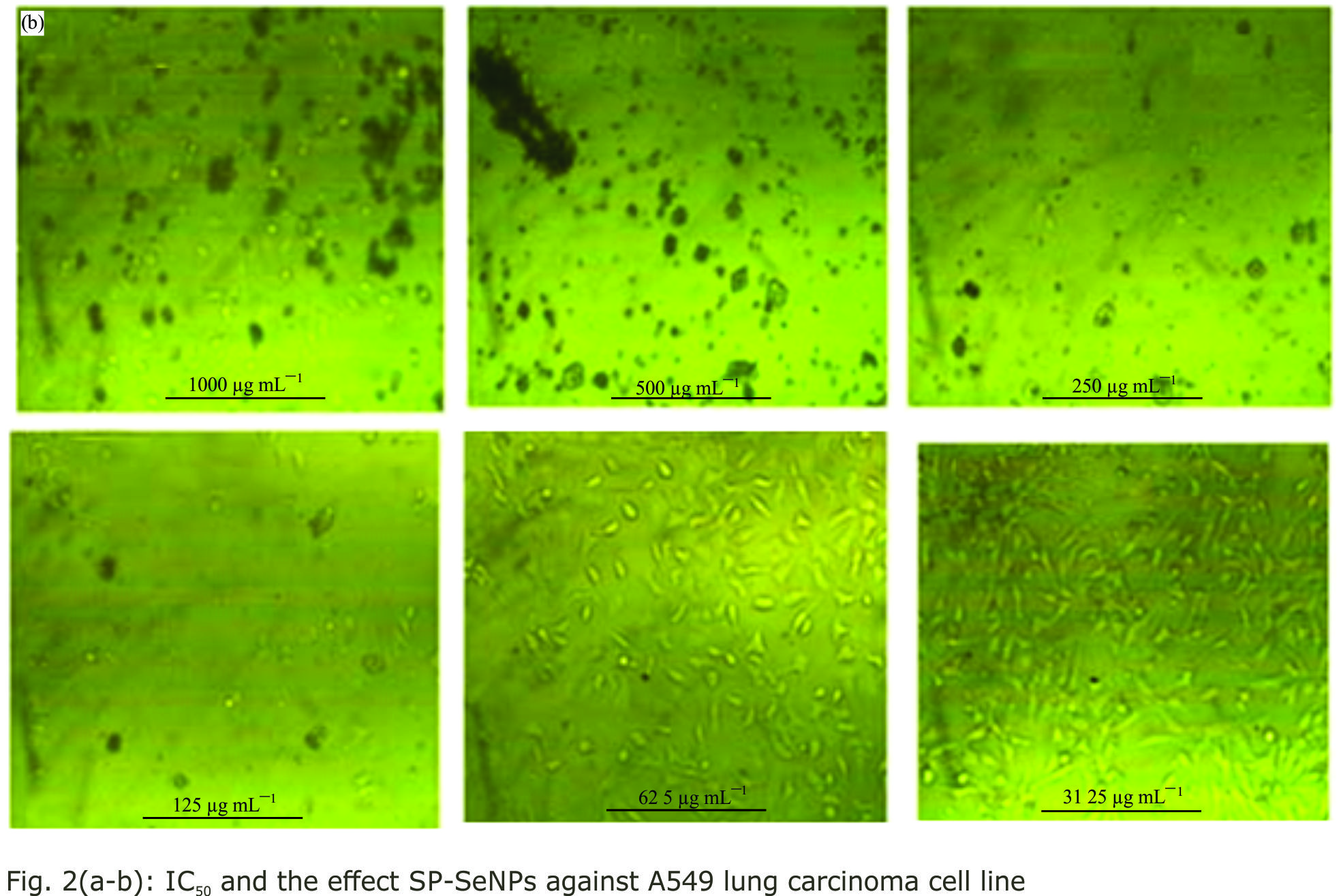
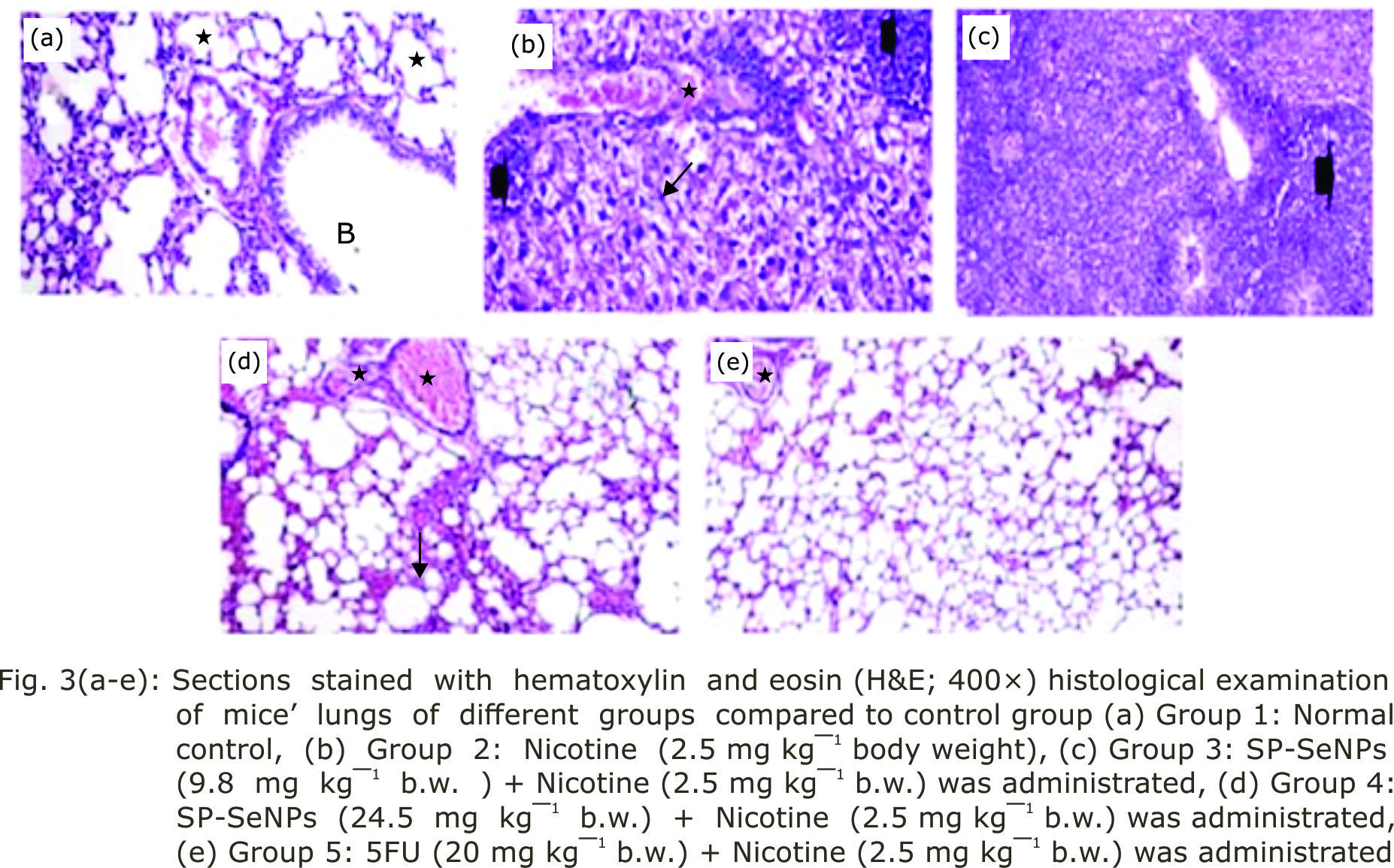
Research Article
Protective Effects of Spirulina Platensis- Selenium Nanoparticles Against Nicotine Induced-Lung Toxicity
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Menna Allah K. Ibrahem
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Amr A. Elgazar
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Hadeer M. Shabib
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Eman A. Morsy
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Sara A. Salah
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Yasmin H. Ibrahem
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Doaa M. Mohamed
Department of Radiology and Medical Imaging, Faculty of Applied Medical science, October 6th University, October 6th City, Egypt
Naglaa A. Gobba
Department of Pharmacology, Faculty of Pharmacy, MUST University, Sixth of October City, Egypt
Mohamed A. Salem
Department of Organic Chemistry, Faculty of Pharmacy, October 6 University, Sixth of October City, Egypt
Fakher M. Ahmed
Department of Organic Chemistry, Faculty of Pharmacy, October 6 University, Sixth of October City, Egypt
Ali A. Ali
Post graduate studies, October 6 University, Sixth of October City, Egypt
Mohammed A. Hussein
Department of Biochemistry, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
LiveDNA: 20.9149