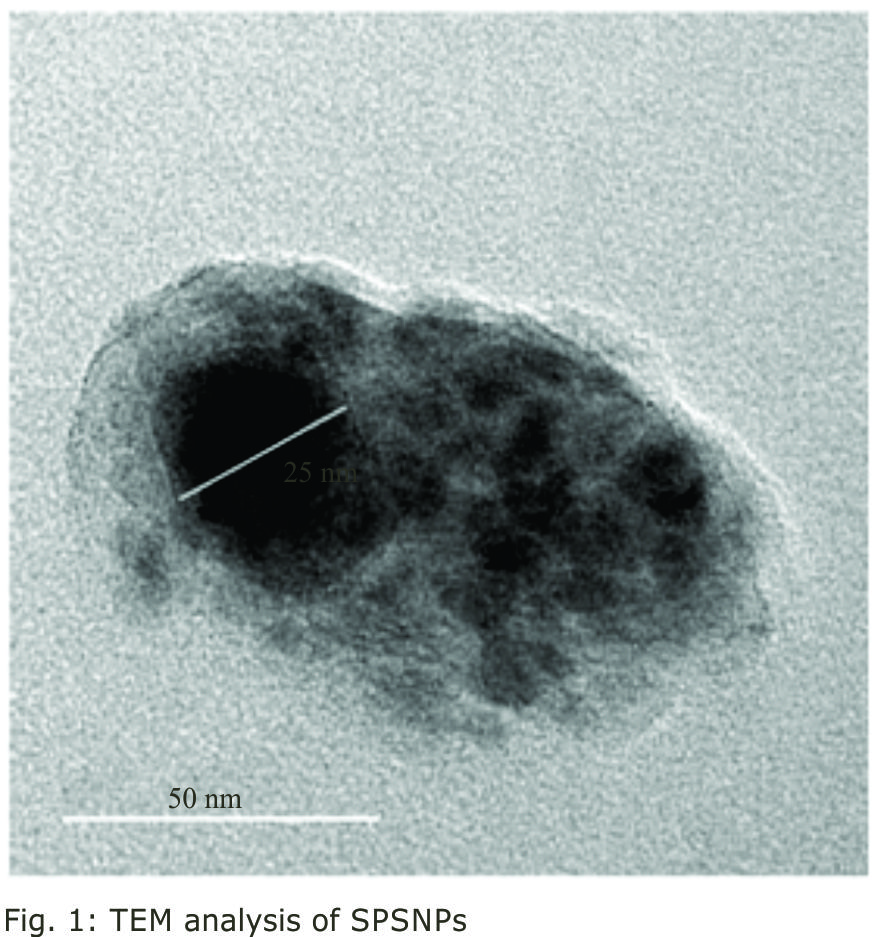
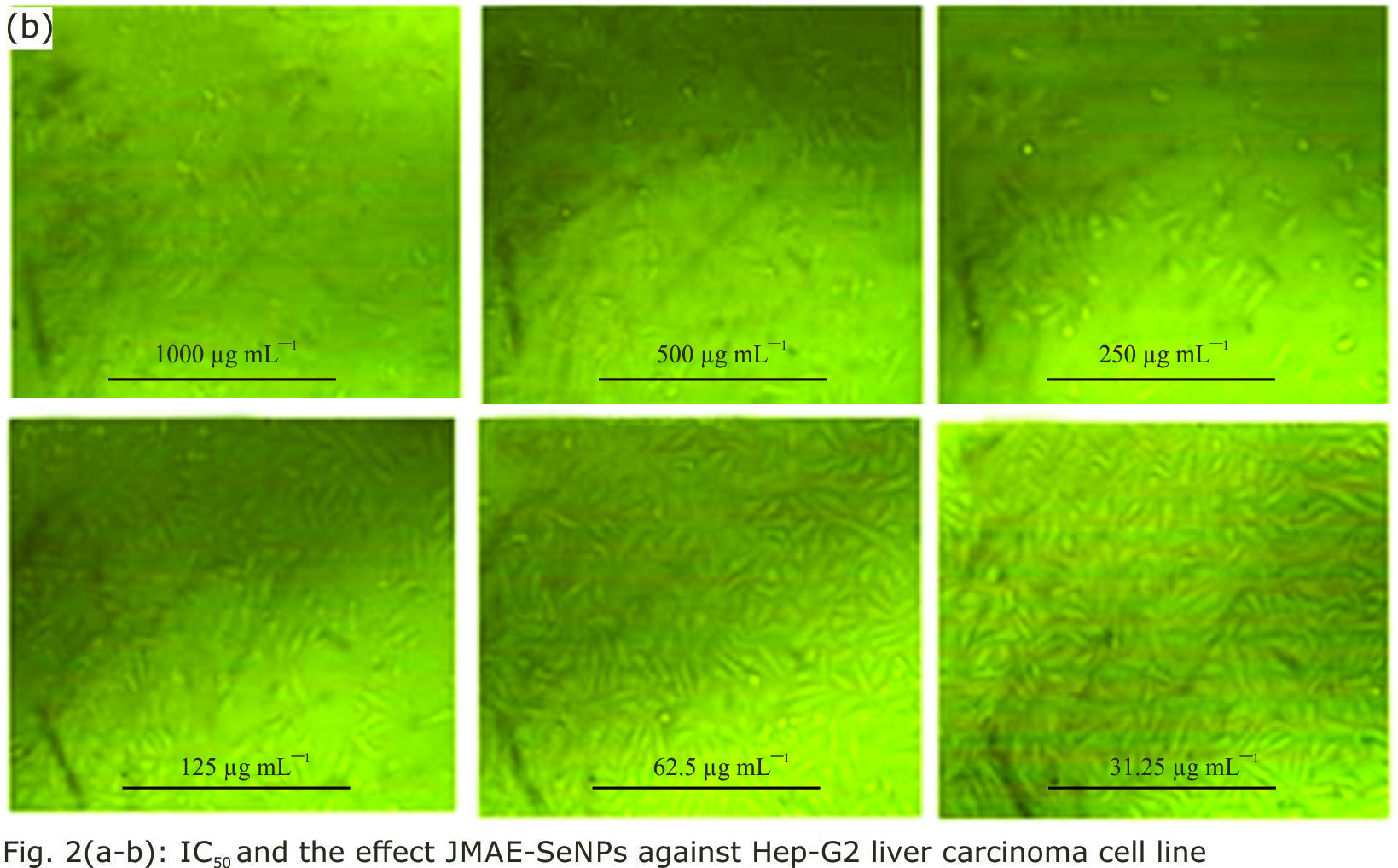
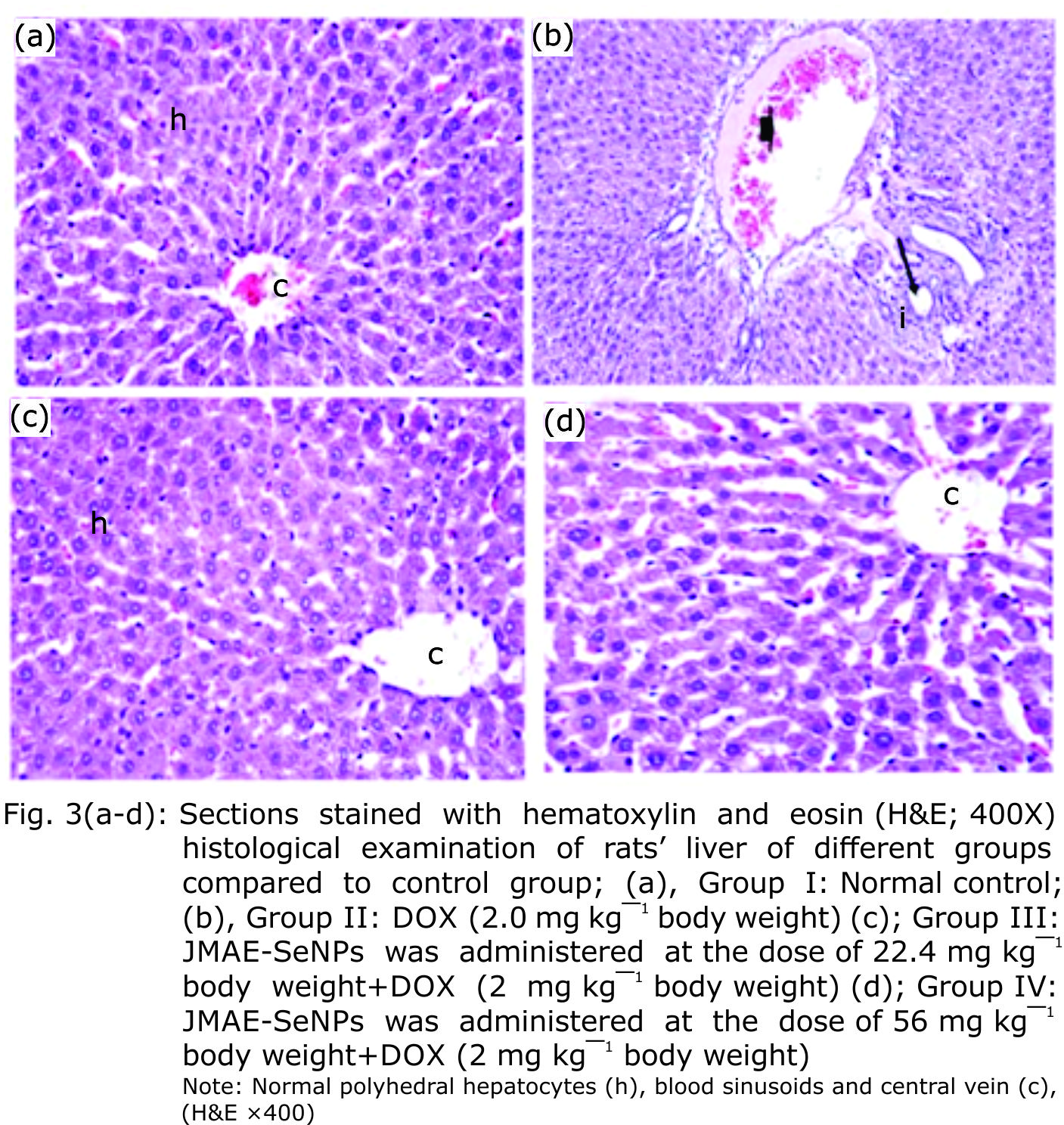
Research Article
Protective Effects of Jasonia Montana-Selenium Nanoparticles Against Doxorubicin-Induced Liver Toxicity
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Dina A. Yousif
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Nada A. Ahmed
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Gehad R. Abd Allah
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Yasmen A. Elbagoury
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Nour E. El Sayed
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Hossam A. Hassan
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Bassem M. El-hefnawy
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Ahmed R. Nageh
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
El-Sayed S. Amer
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Ahmed H. Mohamed
Department of Radiology and Medical Imaging, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
Naglaa A. Gobba
Department of Pharmacology, Faculty of Pharmacy, MUST University, October 6th City, Egypt
Mohammed A. Hussein
Department of Biochemistry, Faculty of Applied Medical Science, October 6th University, October 6th City, Egypt
LiveDNA: 20.9149