Research Article
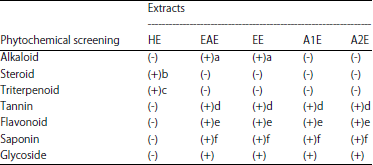
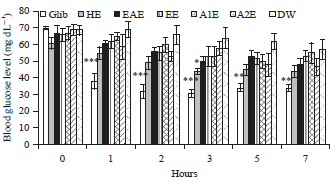
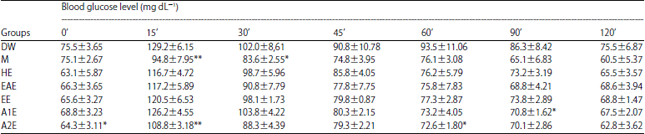
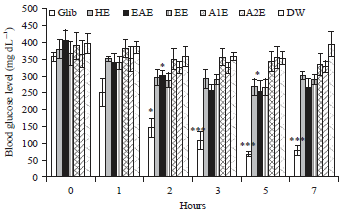
Effect of Lawsonia inermis Linn. Extracts on Blood Glucose Level in Normal and Streptozotocin-Induced Diabetic Rats
Departement of Pharmacology and Therapeutic, Faculty of Medicine, Universitas Sumatera Utara, Medan, 20155, Indonesia
LiveDNA: 62.16072
Y.S. Pane
Departement of Pharmacology and Therapeutic, Faculty of Medicine, Universitas Sumatera Utara, Medan, 20155, Indonesia
N.A. Yusoff
Integrative Medicine Cluster, Advanced Medical and Dental Institute, Universiti Sains Malaysia, Penang, 13200, Malaysia