Research Article
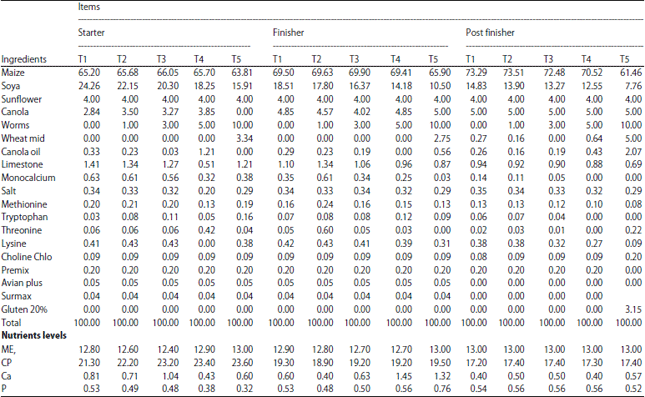
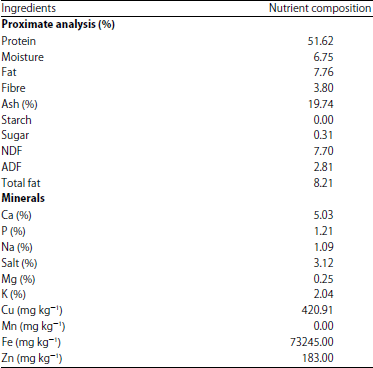
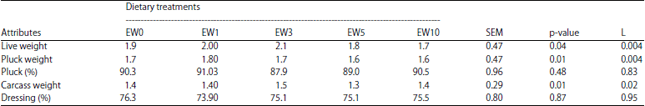
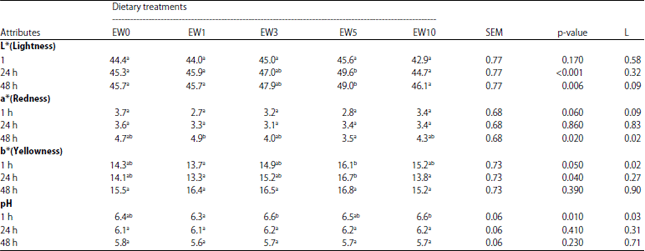
The Effect of Earthworm Eisenia foetida Meal as a Protein Source on Carcass characteristics and Physico-Chemical Attributes of Broilers
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice, 5700, South Africa
LiveDNA: 27.24795
V. Muchenje
Department of Livestock and Pasture Science, University of Fort Hare, Private Bag X1314, Alice, 5700, South Africa
P.J. Masika
Fort Cox College of Agriculture and Forestry, P.O. Box 2187, King Williams Town, 5600, South Africa