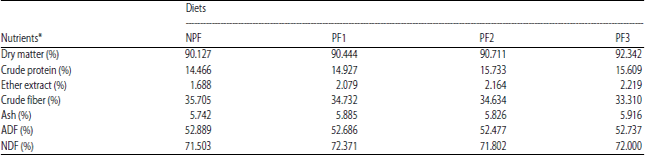
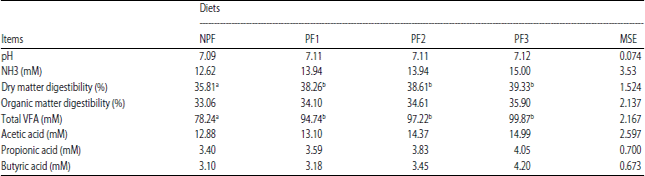
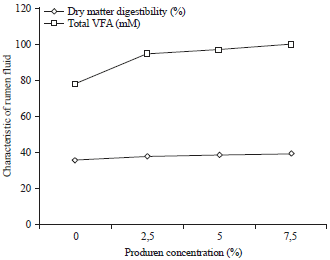
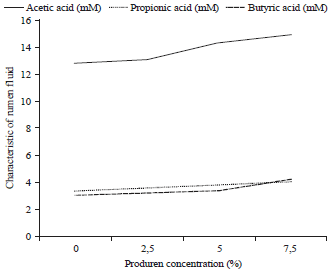
Research Article
In vitro Evaluation of Oil Palm Fronds Fermented with Produren: A Durian Probiotic
Faculty of Animal Husbandry, Jambi University, Kampus Pinang Masak, Jl. Raya Jambi-Ma. Bulian Km. 15. Desa Mendalo Darat, Jambi, Indonesia
E. Musnandar
Faculty of Animal Husbandry, Jambi University, Kampus Pinang Masak, Jl. Raya Jambi-Ma. Bulian Km. 15. Desa Mendalo Darat, Jambi, Indonesia