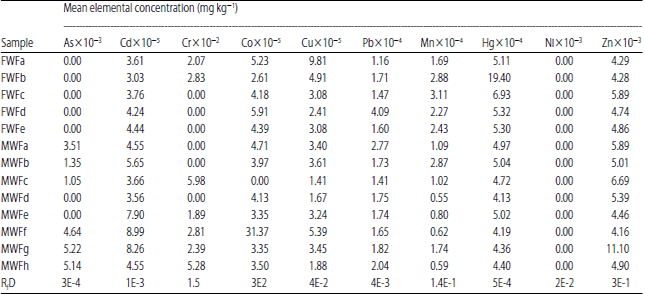
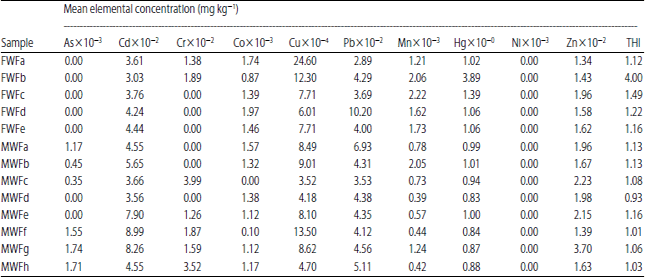
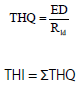Research Article
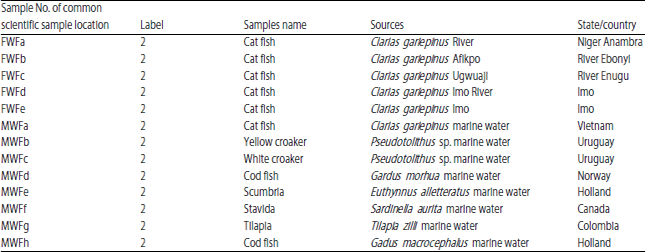
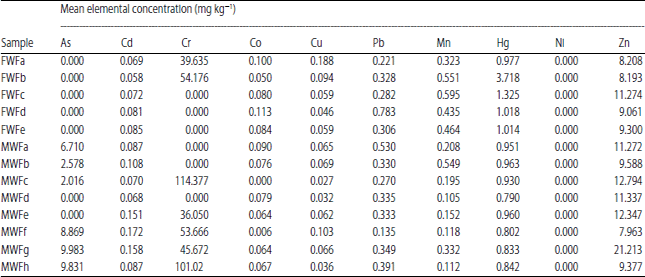
Potential Health Impacts of Heavy Metal Concentrations in Fresh and Marine Water Fishes Consumed in Southeast, Nigeria
Department of Medical Radiography and Radiological Sciences, University of Nigeria, Enugu Campus, Nigeria
C.M.I. Okoye
Department of Physics and Astronomy, University of Nigeria, Nsukka, Nigeria
M.C. Okeji
Department of Medical Radiography and Radiological Sciences, University of Nigeria, Enugu Campus, Nigeria
E.O. Clifford
Department of Physics and Astronomy, University of Nigeria, Nsukka, Nigeria