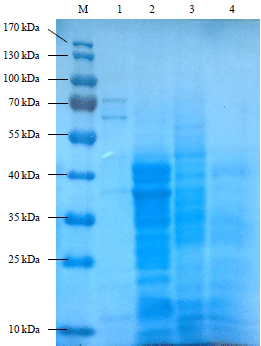
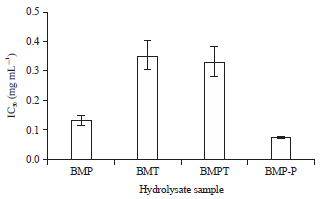
Research Article
Increased Inhibition of Angiotensin Converting Enzyme (ACE) Obtained from Indonesian Buffalo Meat Protein Using SEP-PAK Plus C18
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Jamhari
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Edi Suryanto
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Rusman
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Setiyono
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Endy Triyannanto
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Yuny Erwanto
Department of Animal Product Technology, Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
Ragil Yuliatmo
Department of Leather Processing Technology, Politeknik ATK, Bantul, Yogyakarta, Indonesia