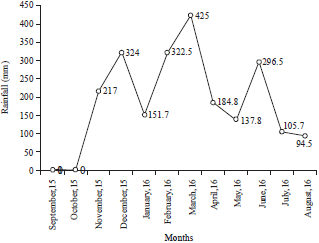
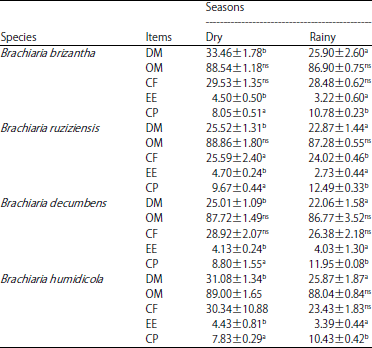
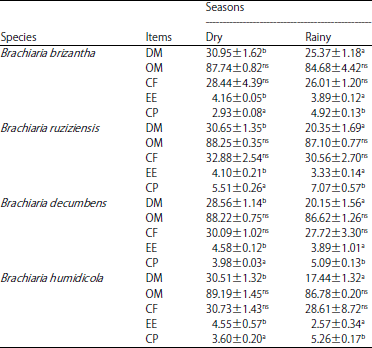
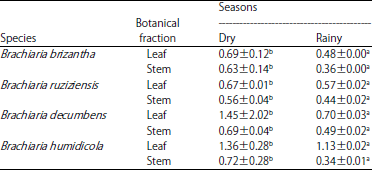
Research Article
Effects of Season, Species and Botanical Fraction on Oxalate Acid in Brachiaria Spp. Grasses in Yogyakarta, Indonesia
Faculty of Animal Science, Gadjah Mada University, Yogyakarta, Indonesia
LiveDNA: 62.23097
Bambang Suhartanto
Faculty of Animal Science, Gadjah Mada University, Yogyakarta, Indonesia
Bambang Suwignyo
Faculty of Animal Science, Gadjah Mada University, Yogyakarta, Indonesia
LiveDNA: 62.14593
Nilo Suseno
Faculty of Animal Science, Gadjah Mada University, Yogyakarta, Indonesia
Farma Herminasari
Faculty of Animal Science, Gadjah Mada University, Yogyakarta, Indonesia