Research Article
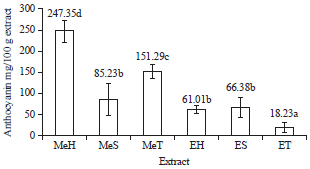
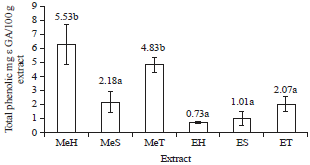
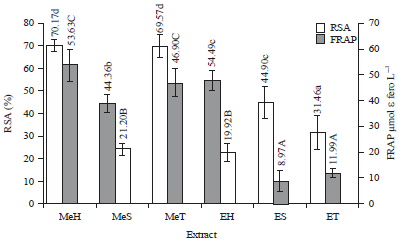
Total Phenolic Content and Antioxidant Activity of Anthocyanin Extract from Purple Yam (Dioscorea alata L.) Flour Using Different Solvents
Department of Food and Agricultural Products Technology, Faculty of Agricultural, Mercu Buana University, Yogyakarta Jl Wates Km 10, 55753 Yogyakarta, Indonesia
LiveDNA: 62.19925
Sri Raharjo
Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Gadjah Mada University, Jl. Flora No. 1, Bulaksumur, 55281 Yogyakarta, Indonesia
Agnes Murdiati
Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Gadjah Mada University, Jl. Flora No. 1, Bulaksumur, 55281 Yogyakarta, Indonesia
Sri Anggrahini
Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Gadjah Mada University, Jl. Flora No. 1, Bulaksumur, 55281 Yogyakarta, Indonesia