Research Article
Quantitative Study of the Effect of Progesterone on the Diabetic Sural Nerves in Rats
Department of Anatomy, Faculty of Medicine, Umm Al-Qura University, Makkah, Saudi Arabia
Human diabetic neuropathy (HDN) occurs in more than 50% of diabetic patients and involves a spectrum of functional and structural changes in peripheral nerves1. Early disorders of nerve function include slowing in nerve conduction velocity (NCV) followed by axonal degeneration, paranodal demyelination and loss of myelinated fibers1-3. Experimental diabetic neuropathy shares a number of features with HDN, such as the structural, functional and biochemical alterations4.
The reduction of Na, K-ATPase activity, together with the decrease in nerve conduction velocity, are the hallmark of diabetic neuropathy5. Increased oxidative stress has been linked to local anesthetic-induced nerve injury in a diabetic neuropathy (DN). Subcutaneous injection of diphenyleneiodonium appears to protect against the functional and neurohistological damage of bupivacaine-blocked sciatic nerves in a high-fat diet/streptozotocin-induced DN model6.
The HDN severely impairs the quality of life because of persistent sensory symptoms, including pain and abnormal thermal perception. At the pathological level, the decrease of intraepidermal nerve fiber (IENF) density in diabetics with symptoms of neuropathy correlates with electrophysiological and psychophysical abnormalities7-10. Diabetic sensory polyneuropathy is characterized by a distal axonopathy of dying-back type11. Previous studies12 described the infiltration and death of monocyte/macrophages and concomitant endoneurial expression of the pro-inflammatory cytokine interleukin-1beta (IL-1beta) and neurotrophic receptor p75 (p75NTR) in the sciatic nerve at the early phases of experimental diabetic neuropathy. The nailfold microcirculation index (MI) is a feasible method for clinical early diagnostic screening of diabetic peripheral neuropathy (DPN) in diabetic patients and is more objective and reliable than the more subjective vibration perception threshold (VPT)13.
Alterations in nociceptive threshold can be reproduced in rats with streptozotocin (STZ)-induced diabetes and can be partially prevented and restored by erythropoietin and prosaposin derived peptide treatments10,11. It was also suggested that miR-199a-3p may have potential as a novel therapeutic target for the treatment of patients with diabetic neuropathy14. Current treatment of diabetic neuropathy relies on the control of glycemic, oxidative stress and neural and vascular risk factors, but this does not fully prevent its occurrence or progression15,16.
Recent findings suggest that neuroactive steroids might exert neuroprotective effects and may be promising in therapy. Laboratories have found that neuroactive steroids themselves and synthetic ligands that interact with their receptors can counteract peripheral nerve degeneration occurring in experimental physical trauma, aging, or in hereditary demyelinating diseases17-20. Similar effects were observed with molecules that induce synthesis in situ of neuroactive steroids (e.g. ligands for peripheral benzodiazepine receptors)21. It was reported that progesterone (P) and its derivatives dihydroprogesterone (DHP) and tetrahydroprogesterone (THP) can stimulate the expression of myelin proteins (i.e., glycoprotein zero, P0 and peripheral myelin protein 22, PMP22)22,23 both in physiological and pathological conditions and preserve the morphology of myelin sheaths24 and axons25.
The aim of the present experiment was to study the morphometric changes after progesterone administration in the diabetic peripheral nerves and evaluate the protecting effects of progesterone against the degenerative changes of diabetes.
Experimental procedures
Animals: A total of 40 two-months-old male Sprague-Dawley rats were used in the present study. Four animals were used as normal control. Diabetes was induced in 36 animals which were divided into two groups. The first group consists of 18 animals which were considered as diabetic control, the second group consisted of 18 animals which received 16 times of injection of progesterone. The animals were handled following the rules approved by Institutional Animal Use and Care Committees.
Induction of diabetes: Diabetes was induced in 36 animals at the same time by a single intravenous injection of freshly prepared streptozotocin (STZ) (65 mg kg–1, Sigma, Milano, Italy) in citrate buffer 0.09 M, pH 4.8 in each animal. Control hyperglycemia was confirmed 48 h after STZ injection and before treatment with steroids by measuring tail vein blood glucose levels using a Glucomen tester. Only animals with mean plasma glucose levels above 300 mg dL–1 were classified as diabetic.
Progesterone injection protocols: Two months after the STZ injection, 18 rats received 16 subcutaneous injections of 1 mg of progesterone (Sigma) dissolved in 200 dL of sesame oil (3.3 mg kg–1 body weight). Injections were administered every 2 days. The rest 18 diabetic rats were used as diabetic nerves. Twenty four hour after the last treatment, the rats were killed and sural nerves were rapidly removed and fixed in 10% formalin.
Morphological analysis: The right and left sural nerves were processed for light microscopic studies. Semi-thin transverse sections (1 m thick) were cut and stained by 0.1% toluidine blue in 1% sodium tetraborate (Merck). Sections were photographed with a Leica I microscope equipped with a Leica DFC320 (Leica, Milano, Italy) camera connected to a Power PC computer, image was acquired with the program Firecam 1.9 (Leica) and processed using Adobe Photoshop 7.1. One semi thin section from each series was randomly selected and used for the morpho-quantitative analysis performed using the freeware program Image J (version 1.39). For the quantitative analysis, a sampling square field, covering an area of 0.003 mm2, was randomly positioned onto the selected digital image. In each sampling field, myelinated nerve fiber profiles were then counted and measured by determining the outer and inner boundary of the myelin sheath using the automatic threshold tool. Therefore, number of fibers, the fiber and axon area and the circle-fitting diameter of fiber (D) and axon (d) were collected. For each sampling region, therefore, calculated the fibers’ density (number per mm2, N mm–2) and the myelin thickness [(D-d)/2].
Statistical analysis: The quantitative data obtained by the experiments had been analyzed through inferential statistical analysis according to the experimental protocols and the nature of the data. Unpaired Student’s t-test was applied to couples of independent variables. Data from experiments with more than two independent variables had been analyzed by one-way analysis of variance (ANOVA) followed by a post hoc comparison test differences of p<0.0001 were considered statistically significant.
Diameter of the myelinated nerve fibers: It was at a mean of 4.171±0.07 μm (ranging from 4.17-4.26 μm) for the control group. The mean was 3.92±0.10 μm (from 3.52-3.92 μm) in the diabetic which is significantly decreased in comparison with the control (p<0.0001). The mean of the diabetic nerves treated with progesterone was 4.11±0.03 μm (ranging from 3.83-4.20 μm). There was a significant increase in the diameter of the myelinated nerve fibers of the treated diabetic nerve than the diabetic (p<0.0001) (Fig. 1, 2).
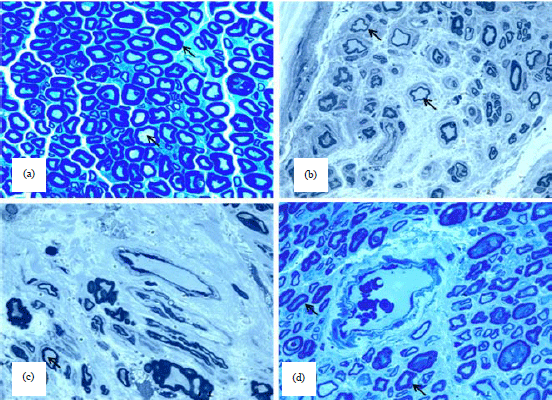 | |
| Fig. 1 (a-d): | (a) Shows the normal compacted myelinated nerve fibers in the endoneurium (arrows), (b, c) Shows the decreased number, diameter and myelin sheath thickness of myelinated nerve fibers in diabetic nerves (arrows) and (d) Shows increased density and myelin thickness with decreased myelin disfigurement of myelinated nerve fibers after progesterone injection. (40X) |
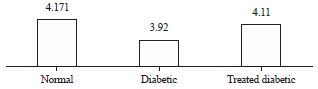 | |
| Fig. 2: | A histogram showing the effect of progesterone administration on the diameters of the myelinated nerve fibers (in μm2) of diabetic nerves (p<0.0001) |
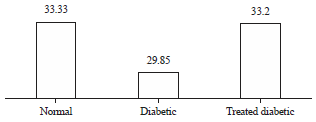 | |
| Fig. 3: | A histogram showing the effect of progesterone administration on the surface areas of the myelinated nerve fibers (in μm2) of diabetic nerves (p<0.0001) |
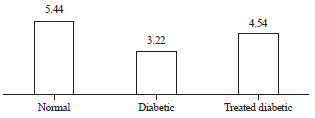 | |
| Fig. 4: | A histogram showing the effect of progesterone administration on the surface areas of the axons of the myelinated nerve fibers (in μm2) of diabetic nerves (p<0.0001) |
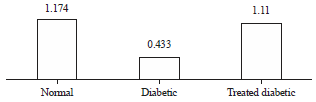 | |
| Fig. 5: | A histogram showing the effect of progesterone administration on the density (number of fibers mm–2) of the myelinated nerve fibers of diabetic nerves (p<0.0001) |
Surface areas of the myelinated nerve fibers: It was at a mean of 33.33±1.42 μm2 (ranging from 30.23-37.54 μm2) in control nerves while that of the diabetic nerves was at a mean of 29.85±0.32 μm2 (ranging from 28.56-38.77 μm2) and showed significant decreased compared to the control group (p<0.0001). The diabetic nerves treated with progesterone injection had a mean of 33.20±1.06 μm2 (ranging from 30.11-37.34 μm2). which was near to the value of the normal nerves.
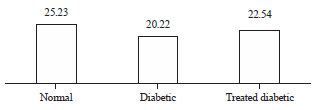 | |
| Fig. 6: | A histogram showing the effect of progesterone administration on the surface areas of the myelin sheaths of the myelinated nerve fibers (in μm2) of diabetic nerves (p<0.0001) |
There was a significant increase in the surface areas of the myelinated nerve fibers of the treated diabetic nerves than the diabetic (p<0.0001) (Fig. 3).
Surface areas of the axons of the myelinated nerve fibers: The surface areas of the axons were at a mean of 5.44±0.19 μm2 (ranging from 4.54-8.01 μm2) for the control group. Those of the diabetic nerves were at a mean of 3.22±0.24 μm2 (ranging from 2.54-6.41 μm2) and diabetic nerves treated with progesterone were at averages of 4.54±0.15 μm2 (ranging from 3.56-8.32 μm2). The axonal areas of the diabetic nerves showed significant reduction compared to the normal (p<0.0001) while the diabetic nerves treated with progesterone showed significant increase in axonal areas than those of the diabetic nerves (p<0.0001). (Fig. 1,4).
Density of the myelinated nerve fibers (mm–2): In the normal control, it was at a mean of 1.174±0.1 mm–2 (ranging from 0.995-1.875 mm–2), in the diabetic nerves, it was at a mean of 0.433±0.04 mm–2 (ranging from 0.321-0.534 mm–2) compared to the density in the diabetic nerves treated with progesterone that was at a mean of 1.11±0.01 mm–2 (ranging from 0.996-1.878 mm–2).
The myelinated nerve fibers density of the diabetic nerves showed significant reduction compared to the control group (p<0.0001). Those of the diabetic nerves which were treated with progesterone showed significant increase in comparison with the diabetic group (p<0.0001) (Fig. 1,5).
Myelin sheath surface areas of the myelinated nerve fibers: The myelin thickness of the myelinated nerve fibers of the control was at a mean of 25.23±0.26 μm2 (ranging from 22.36-29.33 μm2). In the diabetic nerves it was at a mean of 20.22±0.25 μm2 (ranging from 18.66-24.78 μm2) and diabetic nerves treated with progesterone was at a mean of 22.54±0.45 μm2 (ranging from 20.12-24.67 μm2). The myelin sheath surface areas of the diabetic nerves showed significant reduction in comparison with the control group (p<0.0001). The treated diabetic nerves showed significant increase compared to the diabetic nerves (p<0.0001) (Fig. 1,6).
Diabetes mellitus can lead to several complications especially diabetic neuropathy, nephropathy and retinopathy which can lead to sensory/motor loss, renal failure and blindness, respectively26. The earliest complication observed is diabetic polyneuropathy that affects peripheral, sensory, motor and autonomic nerves27. Consequent to neurological complications, diabetic patient often complain of wounds or wound deterioration due to sensory nerve affection or falls accompanied by fractures due to motor nerve function deterioration28. Therefore, protection of diabetic patients against polyneuropathy is vital to avoid further complications29. Myelinated nerve fibers has 10 times nerve conduction velocity than the unmyelinated nerve fibers30.
Schwann cells has a major role in peripheral nerve myelination as these cells produce myelin-associated proteins such as myelin-associated glycoprotein (MAG), myelin basic protein (MBP) and myelin protein zero (P0)31. Immature Schwann cells express high levels of the neurotrophin receptor P75 and low levels of MAG, MBP and P0, whereas mature Schwann cells down-regulate P75 and up-regulate MAG, MBP and P0 as cells become functional myelinating cells31. Schwann cells undergo de-differentiation following injury such as denervation32.
The present study showed that the diameters of the myelinated nerve fibers and axons were significantly decreased in the diabetic nerves. This could be due to decreased vascularity of the endoneurium as a result of affection of the vasa nervosa or decrease in the blood supply to the peripheral nerve27-33. It could be also due to hypoxia, hyerglycemia and increased oxidative stress that contribute directly and indirectly to Schwann cell dysfunction. Consequently, it results in impaired paranodal barrier function, damaged myelin, reduced antioxidative capacity and decreased neurotrophic support for axons4,28-33. It could be also explained by the decrease in the myelin sheath thickness or its abnormal configuration and infoldings as reported during the present study. Progesterone administration showed significant increase in the diameter of the myelinated nerve fibers in the diabetic nerves. The exact mechanism of such improvement is not very clear, however, it was postulated that it reduces myelin infolding4,29-34, reverses impairments in nerve conduction velocity, nociception, skin innervation density and Na+, K+ -ATPase activity, decrease endoneurial edema and also increased mRNA levels of myelin proteins35,36. It was also reported that progesterone and its derivatives administration in ageing peripheral nerves reversed many of the changes seen in the ageing rat nervous system, such as decreased expression of glycoprotein zero and peripheral myelin protein3,36 decreased numbers of thin myelinated fibers, as well as myelin in-folding and irregularities in shape37-39. It can also improve the structural and functional changes in Schwann cells of the diabetic peripheral nerves through reversing the Na+, K+-ATPase activity, decreasing the accumulated endoneurial edema and also through increasing mRNA levels of myelin proteins40,41. Moreover, repeated treatment with the progestin receptor antagonist, mifepristone, caused a decrease in axon diameter and increased neurofilament density in developing peripheral nerves20.
The diameter of the myelinated nerve fibers was significantly increased after administration of the progesterone. This could be probably due to the significant increase in the myelin thickness shown in the current study. Such an increase could be due to stimulation of the expression of myelin proteins (i.e., glycoprotein zero, P0 and peripheral myelin protein 22, PMP22)36,37 by the progesterone. Such stimulation can preserve the morphology of myelin sheaths and myelinated nerve axons21,38 as shown in the present study.
The surface area of the myelinated nerve fibers in the diabetic nerves in the present study showed significant decrease compared to the normal nerves. Such a decrease could be multifactorial as a result of the degenerative changes in Schwann cells, decrease in the thickness of the myelin sheaths, axonal retraction or collapse and the decreased blood supply to the diabetic peripheral nerve. Such a decrease may be partially corrected in some of the myelinated nerve fibers due to the degenerative vacuoles in the cytoplasm of the Schwann cells, fissuring of the myelin sheaths or intra-axonal oedema in the diabetic nerves4,8. It may be also due to the impaired nerve regeneration in diabetes as a result of delayed immediate early gene responses that precede macrophage recruitment as recorded by several authors4,33,35-37. The surface area of the myelinated nerve fibers in the diabetic nerves showed significant increase after progesterone injection which could be attributed to correction of the above pathological derangements in the diabetic nerves.
The number of the myelinated nerve fibers in the diabetic nerves as shown in the current study showed significant reduction compared to the normal. Progesterone treatment of the diabetic nerves on the other hand showed significant increase in the number of myelinated nerve fibers compared to the diabetic nerves. Such decrease in the diabetic nerves could be secondary to the universe ischaemic blood supply to the nerve that resulted in degenerative changes in the whole parts of the nerve17,33. It could be due to the metabolic deficits resulting from metabolic changes in diabetes. These metabolic changes affect the function and structure of Schwann cells leading to appearance of cytoplasmic vacuoles and their degeneration4,6.
The lack of knowledge concerning the mechanisms underlying the actions of progesterone in the nervous system represents a serious limitation on the development of more efficient and targeted treatments of neurological damage and disorders. Progesterone directly regulates gene expression via two intracellular progesterone receptor (PR) isoforms, PR-A and PR-B37,42-45, but their respective roles in mediating the neuroprotective and promyelinating effects of the hormone have not been explored. The PRs interact as dimers not only with DNA progesterone response elements but also with signaling proteins of the Src/Ras/Erk pathway outside the nucleus46,47. The recent identification of membrane receptors of progesterone, unrelated to the intracellular receptors, provides opportunities for the development of new cell membrane-specific progestin ligands48. Major effects of progesterone on neurons and glial cells are mediated by its metabolite allopregnanolone (3a, 5a-Tetrahydro progesterone), which is a potent positive modulator of g-aminobutyric acid type A (GABAA) receptors49. Recently, the antagonism of sigma-1 (s1) receptors by progesterone has been shown to be involved in acute neuroprotection after ischemic brain damage50.
It can be concluded that progesterone has a neuroprotective effects on the diabetic nerves. It corrects the loss, number, diameters, surface areas and myelin sheaths of myelinated nerve fibers. It deserves more clinical trials.