Research Article
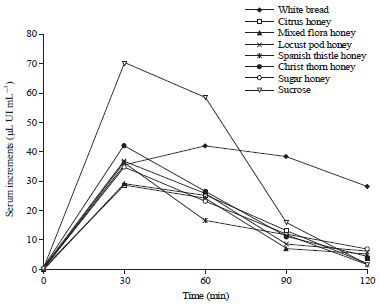
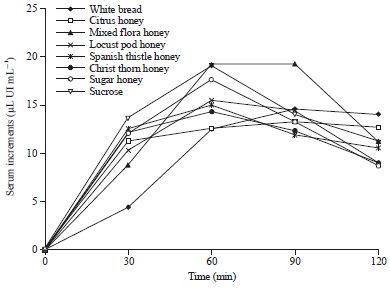
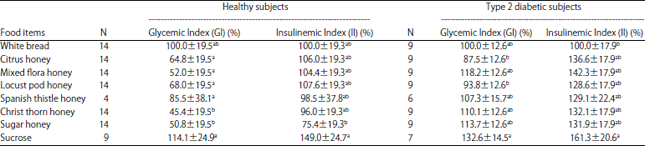
Glycemic and Insulinemic Response of Different Types of Jordanian Honey in Healthy and Type 2 Diabetic Volunteers
Department of Nutrition and Food Technology, Faculty of Agriculture, The University of Jordan, Amman, Jordan
Hamed R.H. Takruri
Department of Nutrition and Food Technology, Faculty of Agriculture, The University of Jordan, Amman, Jordan
LiveDNA: 962.15132
Aly A. Mishal
Department of Endocrinology, Islamic Hospital, Amman, Jordan
Refat A. Alkurd
Department of Nutrition, Faculty of Pharmacy and Medical Sciences, University of Petra, Amman, Jordan