Research Article
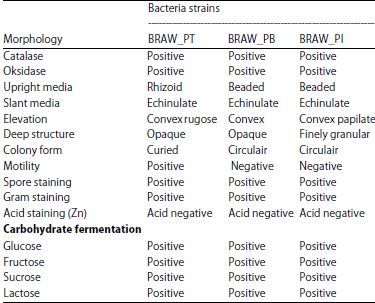
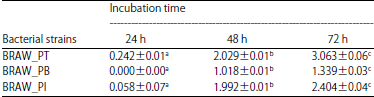
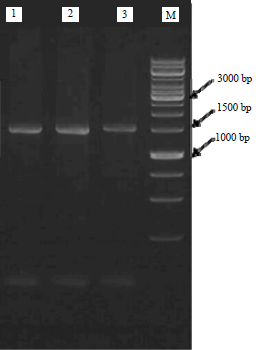
Screening and Characterization of Keratinolytic Bacteria from Puffer Fish Skin Waste
Postgraduate School of Animal Science, Faculty of Animal Science, Universitas Gadjah Mada, Jl. Fauna No. 3, Bulaksumur, 55281 Yogyakarta, Indonesia
Zaenal Bachruddin
Department of Animal Nutrition and Feed Science, Faculty of Animal Science, Universitas Gadjah Mada, Jl. Fauna No. 3, Bulaksumur, 55281 Yogyakarta, Indonesia
Nanung Agus Fitriyanto
Department of Animal Products Technology, Faculty of Animal Science, Universitas Gadjah Mada, Jl. Fauna No. 3, Bulaksumur, 55281 Yogyakarta, Indonesia
LiveDNA: 62.12639
Tomoyuki Nakagawa
Departement of Applied Life Science, Faculty of Applied Biological Sciences, Gifu University, Gifu, Japan
Takashi Hayakawa
Departement of Applied Life Science, Faculty of Applied Biological Sciences, Gifu University, Gifu, Japan
Ambar Pertiwiningrum
Department of Animal Products Technology, Faculty of Animal Science, Universitas Gadjah Mada, Jl. Fauna No. 3, Bulaksumur, 55281 Yogyakarta, Indonesia
LiveDNA: 62.16078