Research Article
Association of the Zinc Content in Hair of School-age Children with the Types of Soil, Age, Sex, Weight and Height in Indonesia
Department of Agritechnology, Faculty of Agriculture, Universitas Borobudur, Jakarta, Indonesia
In the last 20 years, the shortage of zinc (Zn) in the human body continues to increase. Zinc deficiency in humans may interfere with the growth and function of sex and reproduction, taste, smell and appetite1. The most common symptoms include dry and rough skin, dull hair, brittle finger nails, white spots on nails, reduced taste and smell, loss of appetite, mood swings, reduced adaptation to darkness, frequent infections, delayed wound healing, dermatitis and acne2.
Zinc deficiency may suppress child growth, weight loss and fat deposition loss in tissues and it may cause hypogeusia and a lowered immune system3-5. Zinc is needed for cell division and differentiation and is an essential nutrient for normal embryogenesis6. Zinc deficiency was reported in 43% of pregnant women in Nigeria7, a similar study was also conducted by Oguizu8 and the result was that 97.5% of pregnant women were deficient in zinc in Nigeria.
Zinc deficiency is a problem in many communities not only in poor areas but also throughout the world in a mild form9-13. A prevalence of moderate zinc deficiency of 5-30% has been reported in children and adolescents from different countries14. The Zn deficiency produces symptoms such as growth retardation and hypogeusia, if the children are overweight or not well cared for11.
Zinc deficiency in humans is thought to be closely associated with the consumption of foods containing low zinc, metabolic disorders in the body and consumption patterns. Zinc deficiency in consumed food can be caused by such foodstuffs produced from agro-ecological conditions that lack zinc. The contribution of land to animal biology through the ground effect on ecosystems is an important concept in ecology15. Daily intake of zinc is required to maintain a stable state because the human body does not have a special Zn deposit. A high concentration of Zn and Cu has been reported to cause diarrhea and a depressed immune function, growth and reproduction16,17. According to FAO. and WHO.18, the lack of this element may arise either as a result of reduced food intake or decreasing availability of Zn in food.
Research design: A cross-sectional study was conducted in eight villages in the province of West Java and Banten considering different types of soil. The respondents were selected by random sampling of a number of school children who resided in eight villages in the provinces of West Java and Banten, Indonesia. The school children respondents were in the age groups of <6, 6-7, 8-9, 10-11, 12-13 or >13 years. The number of respondents consisted of 329 boys and girls.
Anthropometrics and biochemical data: Furthermore, hair samples were collected from each child, the height and weight were measured and the age and sex were recorded. Hair cutting was performed randomly on the head using a pair of metal-free scissors and then the hair was placed into a plastic bag that had been labeled. The zinc levels were measured using Atomic Absorption Spectroscopy (AAS).
Statistical analysis: The data were processed by the IBM SPSS Statistics 19. The data were analyzed using descriptive statistics (frequency, percentage, mean and standard deviation), cross tabulation and chi-squared analysis were used to identify the types of soil, sex, age, weight and height that have a relationship with the prevalence of zinc deficiency in children. Statistical significance was accepted at a p<0.05.
At the location of the child’s hair sampling in the eight villages, there were only three types of soil: Entisol (Alluvial), Inceptisol (Latosol) and Alfisol (Podsolic). Based on soil chemical analysis, an average availability level of Zn in the soil of that level is 2.65±1.50 ppm, with the range of 0.84-7.72 ppm, as presented in Table 1. Based on the types of soil, the average Zn level of Entisol was less than that of Inceptisol and the average Zn level of Inceptisol was less than that of Alfisol. Furthermore, the Zn content levels available in the soil are classified into three criteria: Low (<0.95 ppm), moderate (0.95-3.10 ppm) and high (>3.10 ppm)19. Based on the criteria of the Zn level in the soil (Fig. 1), the content of Zn in the soil with low criteria involved only Entisol, the Zn level in soil with moderate criteria involved Entisol and was greater than that in Inceptisol, the Zn level in Inceptisol was greater than that in Alfisol and the Zn level in soil with high criteria involved Entisol and was less than that in Inceptisol, the Zn level in Inceptisol was less than that in Alfisol. Thus, the levels of zinc in the soil for the three types of soil are not much different cumulatively for the medium and high criteria.
| Table 1: | Mean, range and criteria of the Zn levels in Entisol, Inceptisol and Alfisol soils |
 | |
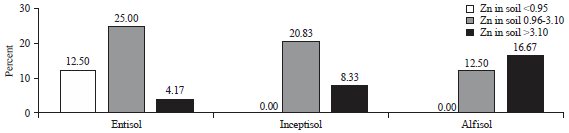 | |
| Fig. 1: | Distribution of the Zn content levels in soil |
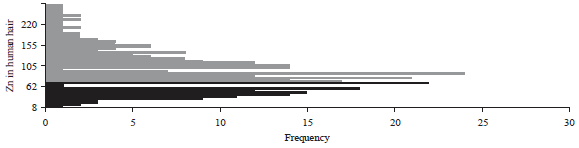 | |
| Fig. 2: | Frequency distribution of the Zn content levels in hair |
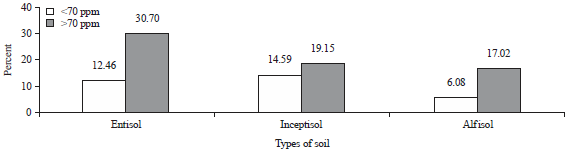 | |
| Fig. 3: | Distribution of the Zn content levels in hair based on the soil type |
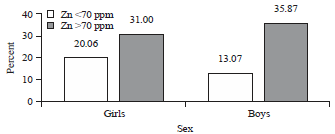 | |
| Fig. 4: | Association of the Zn content levels in hair with sex |
Analysis of the Zn content levels in hair: Based on the analysis of the levels of zinc in hair, 109 children (33.13%) had levels of Zn <70 ppm and 220 children (66.87%) had levels of Zn >70 ppm. In Fig. 2, the frequency distribution of children whose Zn content level in hair was <70 ppm is shown in dark color and that of children whose Zn content level in hair was >70 ppm is shown in grey color.
Association of the types of soil with the levels of Zn in hair: The Zn content levels in hair, in accordance with location of types of soil (Fig. 3), show that the Zn content on Inceptisol is <70 ppm and is greater than Entisol and the Zn content on Entisol is >70 ppm and is greater than Alfisol. Chi-squared test between the Zn levels in hair and soil type showed that there was a significant association a between the soil type and the levels of zinc in hair with a p = 0.019 (<0.05). The Zn content levels of <70 ppm were more common in Inceptisol, Entisol and Alfisol, in that order. Similarly, Zn >70 ppm was more common in Inceptisol, Entisol and Alfisol, in that order.
Association of sex and Zn content levels in hair: Figure 4 shows the Zn content levels in hair for boys and girls were different and there was a tendency that for girls (20.06%), the Zn level in hair <70 ppm was greater than that in boys (13.07%). Similarly, the Zn content levels in the hair >70 ppm were more common in boys than in girls. The results of chi-squared analysis showed that there was a significant association between the levels of zinc in hair and sex (boys and girls) with p = 0.015 (<0.05).
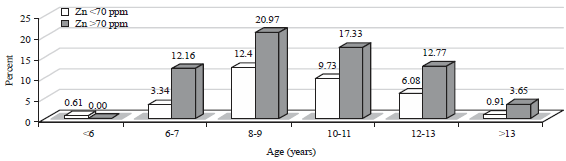 | |
| Fig. 5: | Association of the Zn content levels in hair with age |
 | |
| Fig. 6: | Association of the Zn content levels in hair with height |
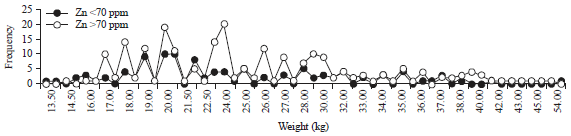 | |
| Fig. 7: | Association of the Zn content levels in hair with weight |
Association between age and Zn content levels in hair: The Zn content levels in hair based on the age of the children are shown in Fig. 5. By age, the levels of zinc in hair was >70 ppm for the age group of 8-11 years and accounted for 126 children (38.30%) and the Zn content levels in hair was <70 ppm for the age group of 8-11 years and accounted for 73 children (22.19%). Children aged less than 11 years old were susceptible to lower Zn content levels in hair, thus, there was a tendency that the concentration of Zn in hair <70 ppm could be reduced with the increasing age of the child. However, chi-squared test between the age and Zn content levels showed no significant association with a p = 0.092 (>0.05).
Association between height (cm) and Zn content levels in hair: In general, the average height of the children was 127±12.77 cm and the mode was 120 cm, the minimum height was 100 cm and the maximum height was 157 cm. Further information about the Zn content levels in hair, based on the height (cm) of the children can be seen in Fig. 6. There was a tendency of the children with a height less than 124 cm to have Zn content levels in hair <70 ppm, a value that is more common than that in children with a height higher than 124 cm. However, based on chi-squared test analysis, no significant association was found between the height and Zn content levels with a p = 0.181 (>0.05).
Association between the weight (kg) and Zn content levels in hair: In general, the average weight of children was 25.42±6.95 kg and the mode was 20 kg, the minimum weight was 13.5 kg and the maximum weight was 54 kg. Further information about the Zn content levels in hair based on the weight of the children is shown in Fig. 7. There was a higher tendency for children who weigh more than 25 kg to have a Zn level in hair <70 ppm than those weighing less than 25 kg. However, the chi-square test analysis showed no significant association between the weight and Zn content levels in hair with a p = 0.291 (>0.05).
Zinc deficiency, both in humans and plants has been known as a critical issue by nutritionists, medical scientists and agronomists for years. It has also been receiving increasing attention recently by other groups, including economists and social scientists, with the recognition that it is a global nutritional problem with significant health, social and economic implications. It has been estimated that one-third of the world’s population is zinc deficient, resulting in numerous health complications, including impairments in the immune system and mental functions. It was also estimated that around half of the world’s agricultural soils are deficient in zinc, leading to decreased crop production and nutritional value. Furthermore, the consumption of cereal-based foods that are typically low in zinc, contributes up to 70% of the daily calorie intake in most of the developing countries, thus resulting in a high prevalence of zinc deficiencies in these populations20. A study performed by Jones15 also found that there were significant differences in the Zn content levels in deer hair in the area being investigated as the reflection of the food of the deer. Zinc is vital for many biological functions in the human body. The adult body contains 2-3 g of zinc, present in all parts of the body, including organs, tissues, bones, fluids and cells. Zinc is essential for the proper functioning of many proteins and more than 100 specific enzymes in the human body21. The zinc content of the major staple foods such as wheat, rice, maize and beans is of particular concern. For many people in developing nations, cereals are the principal source of calories, proteins and minerals. Since these are the regions with widespread zinc-deficient soils, these are also the regions with widespread zinc deficiency in humans. There is a direct and vital link between zinc deficiency in crops and human health in these areas of the world. A study conducted in India documented lower zinc levels (in blood plasma serum) in people feeding on cereal grains with lower zinc content grown in zinc-deficient soils22.
Today, it is estimated that 50% of agricultural soils devoted to cereal cultivation are potentially zinc deficient. Over two-thirds of the rice grown worldwide is produced on flooded paddy soils, which generally contain very low amounts of plant-available zinc. Wheat is typically grown in alkaline and calcareous soils with low organic matter in the semiarid (rain fed) regions of the world. These soil and climactic conditions are likely to make zinc less available for absorption and use by plants. Under zinc-deficient soil conditions, plants show a high susceptibility to environmental stress factors such as drought, heat stress and pathogenic infections, which stimulate the development of chlorosis and necrosis on the leaves and cause stunted growth.
The result of chi-squared test analysis between the Zn content levels in hair and types of soil shows that different types of soil have a significant association with the Zn content levels in hair (p = 0.019). The Zn content levels in hair <70 ppm are commonly found in girls rather than in boys. Furthermore, the chi-squared test result between the Zn content levels in hair and sex showed a significant association (p = 0.015). The analysis by age showed that the Zn content levels in human hair <70 ppm for children aged 8-11 years accounted for 73 children, more than those aged >11 years. However, the chi-square test results showed that age, height and weight had no significant association with the Zn content levels in hair with p = 0.092, 0.181 and 0.291, respectively.
This study evaluated the levels of zinc in soil and in children’s hair. This study showed an association between the levels of Zn in soil with high levels of Zn in the plant as well as in human hair.
The author gratefully acknowledges to several staffs members at the Department of Agritechnology, Faculty of Agriculture, Universitas Borobudur Jakarta for suggestions, guidance and support throughout this study.