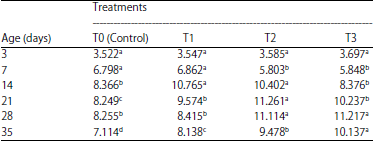
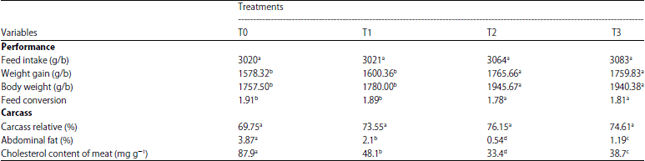
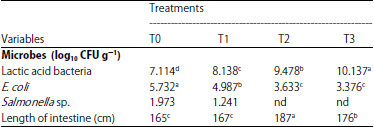Research Article
Impact of Administration Age of Probiotic Lactococcus plantarum on the Intestinal Microflora and Performance of Broilers
Department of Animal Production, Faculty of Animal Science, University of Andalas, Limau Manis Campus, 25163 Padang, Indonesia
Sabrina
Department of Animal Production, Faculty of Animal Science, University of Andalas, Limau Manis Campus, 25163 Padang, Indonesia
F. Arlina
Department of Animal Production, Faculty of Animal Science, University of Andalas, Limau Manis Campus, 25163 Padang, Indonesia
E. Purwati
Department of Animal Production, Faculty of Animal Science, University of Andalas, Limau Manis Campus, 25163 Padang, Indonesia
S.N. Aritonang
Department of Animal Production, Faculty of Animal Science, University of Andalas, Limau Manis Campus, 25163 Padang, Indonesia
H. Abbas
Department of Animal Science, Faculty of Agriculture, University of Taman Siswa, 25111 Padang, Indonesia