Research Article
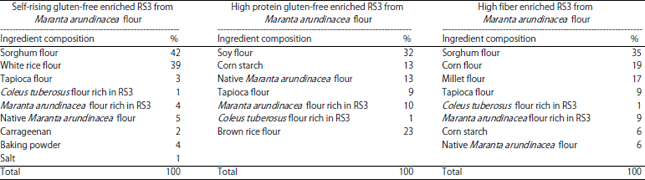
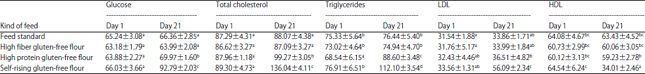
Proximate and Chemical Analysis of Gluten-free Enriched, Resistant Starch Type 3 from Maranta arundinacea Flour and its Potential as a Functional Food
Department of Food Engineering Education, Faculty of Engineering, Yogyakarta State University, Karangmalang, Depok, Sleman 55281 Yogyakarta, Indonesia
Badraningsih Lastariwati
Department of Food Engineering Education, Faculty of Engineering, Yogyakarta State University, Karangmalang, Depok, Sleman 55281 Yogyakarta, Indonesia
Sutriyati Purwanti
Department of Food Engineering Education, Faculty of Engineering, Yogyakarta State University, Karangmalang, Depok, Sleman 55281 Yogyakarta, Indonesia