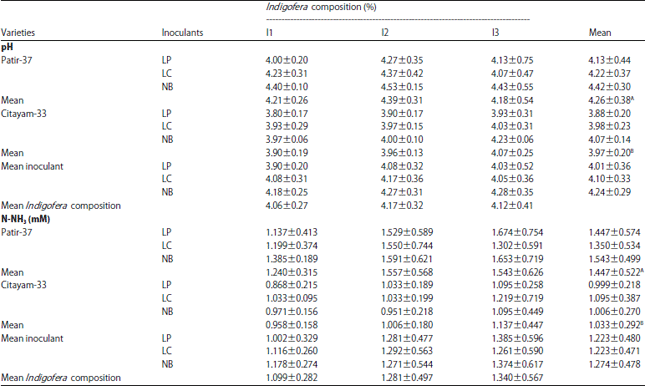
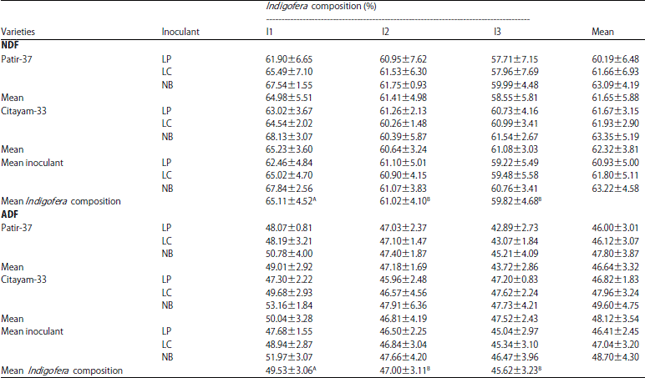
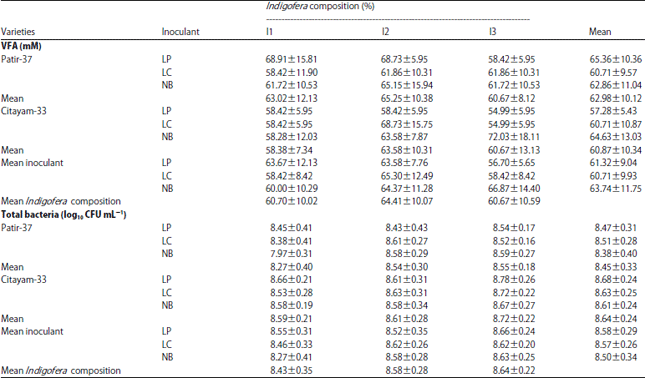
Research Article
Silage Quality of Rations Based on in situ Sorghum-Indigofera
Department of Feed and Nutrition, Bogor Agricultural University, Indonesia
K.G. Wiryawan
Department of Nutrition Science and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
P.D.M.H. Karti
Department of Nutrition Science and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
I.G. Permana
Department of Nutrition Science and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia
L. Abdullah
Department of Nutrition Science and Feed Technology, Faculty of Animal Science, Bogor Agricultural University, Indonesia