Research Article
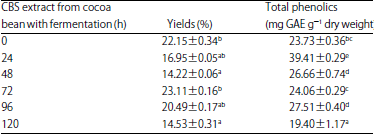
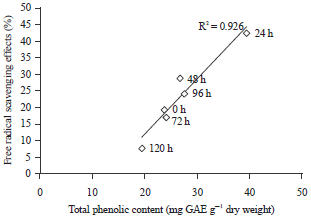
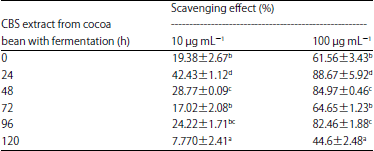
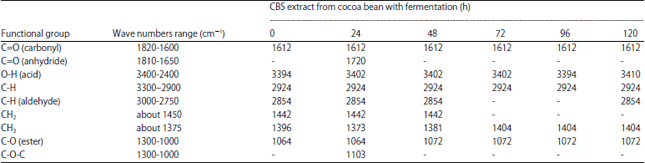
Effects of Cocoa Bean (Theobroma cacao L.) Fermentation on Phenolic Content, Antioxidant Activity and Functional Group of Cocoa Bean Shell
Center for Plantation-Based Industry, Ministry of Industry, 90231 South Sulawesi, Indonesia
R. Armunanto
Department of Chemistry, Faculty of Mathematics and Natural Science, Gadjah Mada University, Sekip Utara, 55281 Yogyakarta, Indonesia
S. Rahardjo
Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Gadjah Mada University, Jl. Flora No. 1, Bulaksumur, 55281 Yogyakarta, Indonesia
Supriyanto
Department of Food and Agricultural Product Technology, Faculty of Agricultural Technology, Gadjah Mada University, Jl. Flora No. 1, Bulaksumur, 55281 Yogyakarta, Indonesia