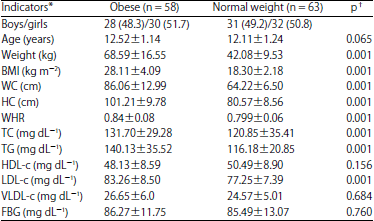
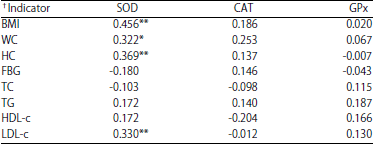
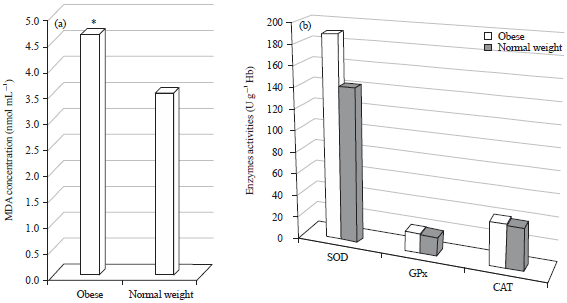
Research Article
Evaluation of Oxidant-antioxidant Status in Obese Children and Adolescents
Department of Nutrition and Food Technology, Faculty of Agriculture, University of Jordan, 11942 Amman, Jordan
Hayder A. Al-Domi
Department of Nutrition and Food Technology, Faculty of Agriculture, University of Jordan, 11942 Amman, Jordan