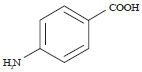
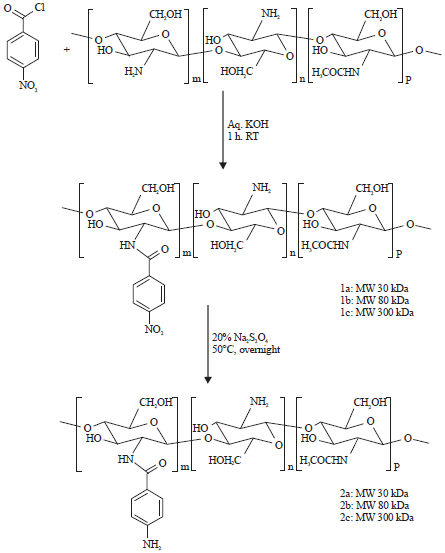
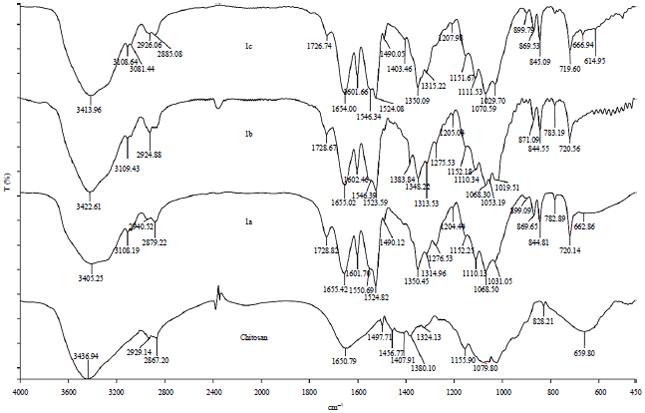
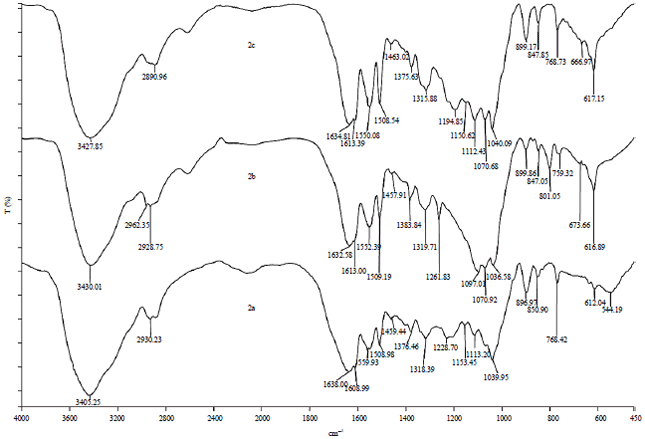
Research Article
p-Aminobenzoic Acid-chitosan Conjugates for PABA Delivery to the Large Intestine
Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat Yai, 90112 Songkhla, Thailand
Chitchamai Ovatlarnporn
Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Prince of Songkla University, Hat Yai, 90112 Songkhla, Thailand