Research Article
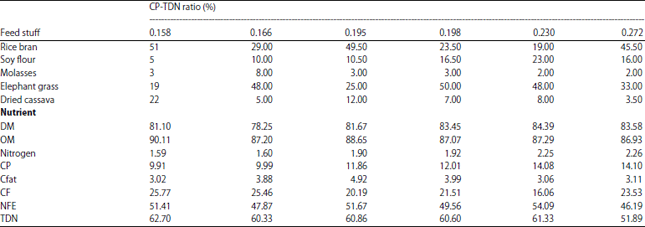
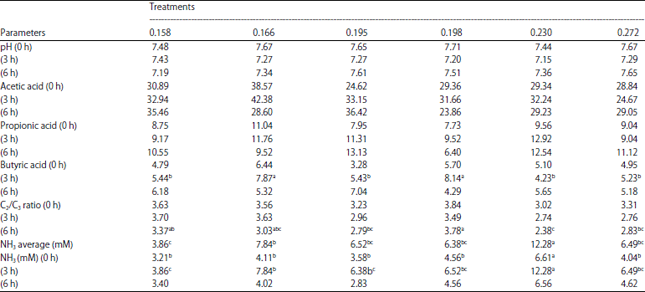
Effect of Feed with Different Energy-protein Ratios on Parameters of Sheep Ruminal Fermentation
Department of Animal Science and Agriculture, University of Diponegoro, Semarang, Central Java, Indonesia
Agung Purnomo Adi
Department of Animal Science and Agriculture, University of Diponegoro, Semarang, Central Java, Indonesia
Soedarsono
Department of Animal Science and Agriculture, University of Diponegoro, Semarang, Central Java, Indonesia
Sunarso
Department of Animal Science and Agriculture, University of Diponegoro, Semarang, Central Java, Indonesia