ABSTRACT
Effect of treatment conditions [soaking, boiling and fermentation with Rhizopus oligosporus (SBF)] on the water soluble vitamins and trypsin inhibitor of bambara nut flour [Vigna subterranea (L) Verdc] was investigated. It was observed that the thiamine content decreased by 5%, riboflavin 6.2%, folacin 9.6%, niacin 10.2% and biotin 14% after 24 h of soaking at room temperature. Treatment by boiling for 45 min revealed that there was a further decrease of 52.4%, 56.2%, 35.0%, 70.0% and 48.3% in the values of thiamine, riboflavin, folacin, niacin and biotin respectively. However, fermentation with Rhizopus oligosporus for 24 h significantly reduced the thiamine content from the original value of 0.47+0.13-0.19+0.02 mg/100g, while riboflavin content increased significantly from 0.15+0.06-0.17+0.01 mg/100 g, folacin 0.16+0.02-0.23+0.03 mg/100 g, niacin 1.88+0.09-2.16±0.00 mg/100 g and biotin 0.10+0.02-0.16±0.05 mg/100 g (P < 0.05). Trypsin Inhibitor Activity (TIA) decreased by 22.1% after soaking for 24 h at room temperature and further decreased by 72% after boiling for 45 mins. Conversely, during fermentation period of 24 h, the TIA increased significantly from 0.71+0.62-1.33±0.59 mg/100 g protein (P < 0.05).
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjn.2009.835.840
URL: https://scialert.net/abstract/?doi=pjn.2009.835.840
INTRODUCTION
Vitamins are organic nutrients found in natural foods which are essential in minute quantities to the nutrition of animals (NSTA, 2001). Vitamins act especially as coenzymes and precursors of coenzymes in the regulation of metabolic processes but do not provide energy or serve as building units (Keuth and Bispring, 1993). Uzogara et al. (1992) reported that legumes are excellent sources of thiamine, folic acid, niacin, riboflavin and biotin, while Minussi et al. (2003); Zhang and Hamauzu (2004) confirmed that fruits and vegetables are good sources of natural vitamins. The deficiency of these vitamins in diets can lead to symptoms such as constipation, appetite, cardiovascular and muscular defects (Alexander, 1995). Severe thiamine deficiency disease known as beriberi is the result of a diet which is carbohydrate rich and thiamine deficient (http://web-instate.edu/theme/mwking/vitamin.htm/). Thiamine is necessary as a co-factor for the pyruvate alongside with μ-ketoglutarate dehydrogenase catalysed reaction as well as the trans-ketolase catalysed reaction of the pentose phosphate pathway
(http://www.file://A:/vitamin-1-watersolublevitamin.htm) while folacin plays a role in nucleic acid biosynthesis which is essential for normal proliferation and functions of the cells.
Niacin which is also known as vitamin B3 nicotinic acid and nicotinamide are both used by the body to form coenzyme Nicotinamide Adenine Dinucleotide (NAD) and Nicotinamide Adenine Dinucleotide Phosphate (NADP) which are the active forms of niacin (http://www.anyvitamins.com/vitambz-niaicnc-into.htm,) Niacin facilitates lipid catabolism and also plays a key role in TCA cycle. Lack of niacin causes the deficiency syndrome pellagra which is characterized by weight loss, digestive disorder, depression and dermatitis (http://www.anyvitamins.com/vitambz-niacinc-into.htm).
Biotin generally classified as B complex vitamins is synthesized by bacteria, yeast, moulds, algae and some plant species (Hidgon, 2004). Deficiencies are seen after a prolonged antibiotic therapy which deplete the intestinal fauna (http://www.file://A:/vitamins-1-watersolublevitamins.htm). Dry skin, fungal infections rashes, fine and brittle hair, changes in mental status and generalized muscular pains are deficiency symptoms of biotin.
Riboflavin is a water soluble B-complex vitamin which is primarily found as an integral component of the coenzyme Flavine Adenine Dinucleotide (FAD) and Flavine Mononucleotide (FMN) (http://www.hoptechno.com/book29.htm). Riboflavin plays a critical role in the body’s energy production. When active it takes the form of FAD or FMN which has the ability to attach to protein enzymes and allow oxygen-based energy production to occur (http://www.whfoods.com/whoeverar.php). A deficiency of riboflavin is characterized by weakness, sore throat, hyperemia, edema of the pharyngeal, mucous membrane, angular stomatitis, corneal visualization and anaemia associated with red hypoplasma of the bone marrow (http://www.indstate.edu/theme/mwking/vitamins.htm).
In general, cooking time, soaking and fermentation period were reported to have pronounced effects on the vitamin levels and anti-nutritional factors present in natural foods (Chavan and Kadam, 1989; Kaack, 1994).
Bambara groundnut [Vigna subterranean (L) Verdc] is an indigenous African grain legume (Poulter, 1981). The crop is reputable as a ‘poorman’s crop’ coupled with high nutritional potentials (Poulter, 1981). It is reported to possess a gross energy value greater than those of other common pulses such as cowpeas, lentil and pigeon pea. It is underutilized in sub-sahara Africa (FAO, 1982). The seed was reported to contain 63% carbohydrate, 19% protein and 6.5% oil (Karikari, 1971). Dried bambara nut can be processed into fried bean cake (akara) or steamed beans paste (moin-moin) (Obizoba, 1993). Milk produced from bambara nut was reported to compare favourably in flavour and composition with cowpea and soybean milk (Poulter, 1981). Despite all its nutritional potentials, there is scanty information on the effect of treatment conditions on water soluble vitamins and trypsin inhibitor activity (TIA) present in the pulse.
Therefore, studies were undertaken to provide information on the effect of treatment conditions [soaking, boiling and fermentation (SBF)] on water soluble vitamins and TIA of bambara nut flour.
MATERIALS AND METHODS
Preparation of Inoculum: Rhizopus oligosporus strain NRRL 2710 was obtained from Department of Biological Sciences University of Agriculture, Abeokuta, Nigeria. The organism was cultured and sub-cultured on Potato Dextrose Agar (PDA) and finally transferred to a slant in McCarthany bottles containing 4% malt extract, 0.5% peptone and 1.5% agar with pH adjusted to 4. It was incubated for 5 days at 30oC for sporulation (Ashenafi and Busse, 1991). 10ml of sterile distilled water was added to the slant and shaken vigorously to dislodge the spores. 1ml of spores suspension was taken and transferred into a haemocytometer for spores enumeration.
Preparation of bambara nut flour: 1.5kg of matured Bambara nut was cleaned properly in water and soaked in clean water (1:6 Bambara nut to water w/w) at room temperature for 24 hours (Prinyawiwatkul et al., 1997). The soaked nuts were boiled and soaked water for 45 minutes, drained and cooled to 25-30oC. 200g portion of the boiled nuts was oven dried at 55oC for 24hrs and stored in a sterile polyethylene bag. The remaining boiled seeds were allowed to drain and cool to room temperature. This was divided into five equal portions of 200g each. These portions were inoculated with 2ml of the spore suspension of Rhizopus oligosporus (1.8’ 106 cfu/ml). The inoculated nuts were placed in perforated plastic bags and placed on a wire mesh screen and fermented at 30oC for 0, 15, 18, 21, and 24 hours respectively. Fermented nuts were later dried in oven at 55oC for 24hours and finely grinded into flour.
 |
Determination of trypsin inhibitor activity (TIA) and water soluble vitamins: Trypsin inhibitor activity was determined in triplicates by the Kakade enzymic assay (Kakade et al., 1974) and trypsin inhibitor units were calculated according to Gaborit et al. (1993), where one trypsin inhibitor unit (TIU) is defined as a decrease of 0.01 absorbance units at 410nm per 10ml assay solution.
Determinations of thiamine, biotin, niacin, riboflavin and folacin were carried out using combined methods of the Association of Vitamin Chemists (1966) and Association of Official Analytical Chemists (2003). All treatments were carried out in triplicates.
RESULTS
The effects of soaking, boiling and fermentation with R. oligosporus on water soluble vitamins and trypsin inhibitor activity (TIA) of bambara nut flour were investigated. It was observed that during soaking of bambara nut for 24hours at room temperature, the thiamine content decreased significantly from the original value of 0.47±0.13mg/100g to 0.446±0.05mg/100g and this further decreased to 0.23±0.03 mg/100g after 45mins of boiling. However inoculation with Rhizopus oligosporus spores initiated a significant decrease from 0.23±0.03 mg/100g to 0.19±0.02mg/100g after 24h of fermentation period at (P<0.05) (Fig. 1).
The riboflavin content of bambara nut also decreased significantly from 0.155±0.06 to 0.145±0.02mg/100g during soaking for 24h at room temperature (P<0.05). A further decrease to 0.064±0.16mg/100g was observed after boiling for 45mins while during fermentation period of 24h the riboflavin content increased sharply to 1.687±0.01mg/100g (Fig. 2).
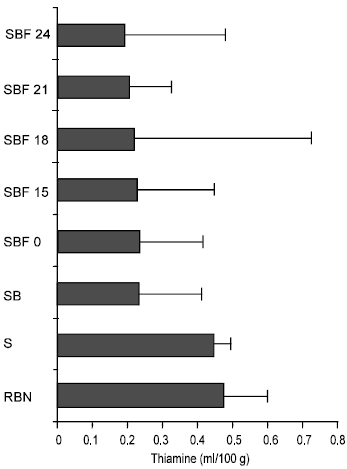 | |
| Fig. 1: | Effect of Soaking, boiling and fermentation on the Thiamine content of Bambara nut |
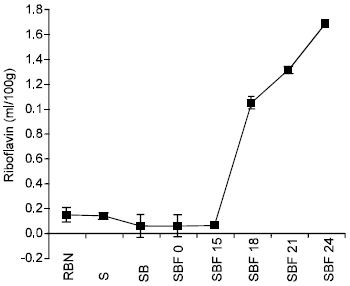 | |
| Fig. 2: | Effect of Soaking, boiling and fermentation on the Riboflavin content of Bambara nut. |
During soaking for 24h at room temperature the original niacin value of raw bambara nut decreased significantly from 1.88±0.09mg/100g to 1.69±0.34mg/100g while boiling for 45mins further decreased the value to 0.564±0.20mg/100g, but fermentation for 24h increased the value significantly from 0.564±0.20mg/100g to 2.162±0.00mg/100g (P<0.05) (Fig. 3).
The effect of soaking the beans for 24hours at room temperature revealed a decrease from 0.10±0.02mg/100g to 0.086±0.05mg/100g while the combined effect of soaking and boiling resulted in a further decrease from 0.086±0.05mg/100g to 0.048±0.07mg/100g. Contrarily, fermentation period of 24h significantly increased the biotin content from 0.048±0.07mg/100g to 0.16±0.05mg/100g (P<0.05) (Fig. 4).
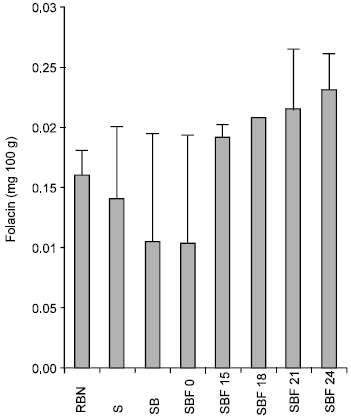
Fig. 5 shows the result of the three treatments on the folacin status of the beans. Soaking only for 24h at room temperature produced a decrease from 0.16±0.02mg/100g to 0.14±0.06mg/100g while boiling for 45mins led to a decrease from 0.14±0.06mg/100g to 0.10±0.09mg/100g which later increased to 0.23±0.03mg/100g after 24h of fermentation.
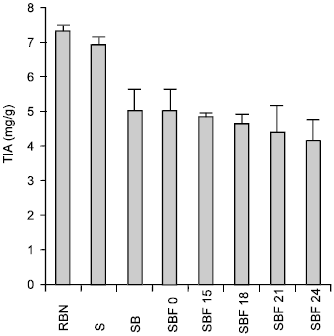
The original TIA of raw bambara nut was seen to decline from 7.32±0.15mg/g protein to 5.11±0.23mg/g protein after soaking for 24h at room temperature (Table 1). During boiling for 45mins the TIA further decreased from 5.11±0.23mg/g protein to 0.71±0.00mg/g protein and then increased significantly to 1.33±0.38mg/g protein after 24h of fermentation.
DISCUSSION
The effects of soaking, boiling and fermentation with Rhizopus oligosporus on the water soluble vitamins content of bambara nut flour were investigated. Thiamine content decreased during soaking by 19.2%, this result is in agreement with the earlier observations of Prinyawiwatkul et al. (1997) during studies on cowpeas. Boiling of the flour for 45 minutes led to a further decrease in thiamine content. This result is comparable with previous studies reported by Waters et al. (1993). Similarly, fermentation of the flour with R. oligosporus for 24h showed that there is a significant reduction in thiamine content of the flour. This observation is similar to previous results reported by Chavan and Kadam (1989) on reduction in thiamine content of rice fermented with R. oligosporus. However, Philips et al. (1983) also reported decrease in thiamine content of fermented cowpea while Wang and Hesseltine (1966); Murata et al. (1967); Van Veen et al. (1970) and Keuth and Bispring reported a decrease in thiamine content of fermented wheat. This decrease in thiamine content may be due to rapid utilization of thiamine for optimum growth and other functions at a higher rate than synthesis of thiamine by the fermenting organism (Wang and Hesseltine, 1966).
Similarly, riboflavin and biotin content of Bambara nut flour decreased after 24hours of soaking. This result conforms with the observations of Phillips et al. (1983) by reporting a similar trend during soaking of cowpea for 24hr. Boiling further enhanced decrease in riboflavin and biotin content. Similar observations were reported by Phillips et al. (1983) and Uzogara et al. (1992) for boiled cowpea, while Deosthale (1979) observed such trend in riboflavin content of chicken peas and green peas. Conversely, during 24hours fermentation, riboflavin and biotin content of the Bambara nuts flour were observed to increase. Phillips et al. (1983) reported a similar result for cowpea.
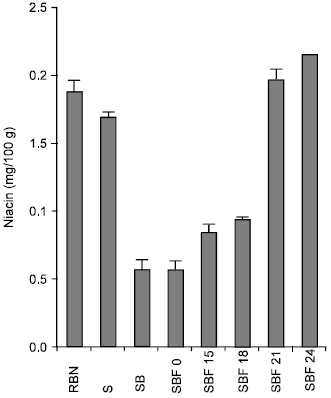 | |
| Fig. 3: | Effect of Soaking, boiling and fermentation on the Niacin content of Bambara nut |
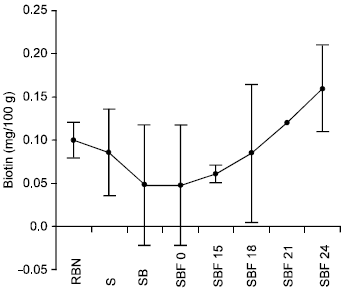 | |
| Fig. 4: | Effect of Soaking, boiling and fermentation on the biotin content of Bambara nut |
Folacin and niacin contents of the flour decreased during soaking for 24h this is also in agreement with previous studies reported by Prinyawiwatkul et al. (1997) on cowpea.
 | |
| Fig. 5: | Effect of Soaking, boiling and fermentation on the Folacin content of Bambara nut. |
 | |
| Fig. 6: | Effect of Soaking, boiling and fermentation on the TIA content of Bambara nut |
Boiling of the nuts led to a significant reduction in folacin and niacin content of the flour; this finding corroborates the submissions of Murata et al. (1967) on fermentation of soybeans. The observed increase in water soluble vitamins with the exception of thiamine may probably be due to synthesis of these vitamins by the mould.
| Table 1: | Results of Trypsin Inhibitor Activity |
 | |
The effects of soaking, boiling and fermentation on the Trypsin Inhibitor Activity (TIA) of Bambara nut were also investigated. The TIA decreased after soaking for 24h. This observation conforms with the earlier report of Havala (1995) who noted that soaking of mug beans, lentils, chickpeas and broad beans for 24h removed 66%, 93%, 50% and 100% of their initial TIA content respectively. Ologhobo and Fetuga, (1984); Gatta et al. (1989) also reported similar results for some cultivars of cowpeas. Furthermore, a significant decrease in TIA content of flour is recorded after boiling the nuts for 45 minutes prior to milling. Chung et al. (1981) reported similar results in their previous studies. Ayagari et al. (1980) stated that boiling destroys TIA in cereal and leguminous grains thereby enhancing its protein digestibility.
There was a slight increase in TIA after 15 hours fermentation of the nuts. This observation conforms with the earlier reports of Prinyawiwatkul et al. (1997) for cowpea flour fermentation. Wang and Hesseltine (1972) reported that increase in TIA of cereal/leguminous grains during fermentation might be due to release of heat resistant bound TIA by protease enzymes produced by R. oligosporus.
REFERENCES
- Kakade, M.L., D.E. Hoffa and I.E. Liener, 1973. Contribution of trypsin inhibitors to the deleterious effects of unheated soybeans fed to rats. J. Nutr., 103: 1772-1778.
Direct Link - Minussi, R.C., M. Rossi, L. Bologna, L. Cordi, D. Rotilio, G.M. Pastore and N. Duran, 2003. Phenolic compounds and total antioxidant potential of commercial wines. Food Chem., 82: 409-416.
CrossRefDirect Link - Murata, K., H. Ikehata and T. Miyamoto, 1967. Studies on the nutritional value of tempeh. J. Food Sci., 32: 580-586.
CrossRefDirect Link - Obizoba, I.C., 1993. Effects of germination, dehulling and cooking on the nutritive value of cowpea (Vigna unguiculata) flour. J. Food Sci., 54: 1371-1372.
Direct Link - Ologhobo, A.D. and L.F. Babatunde, 1984. The effect of processing on the trypsin inhibitor, hemagglutinin, tannic acid and phytic acid contents of seeds of ten cowpea varieties. J. Food Process Preser., 8: 31-40.
CrossRef - Phillips, R.D., M.S. Chinnan, A.L. Branch, J. Miller and K.H. McWatters, 1988. Effects of Pretreatment on functional and nutritional properties of cowpea meal. J. Food Sci., 53: 805-809.
CrossRefDirect Link - Poulter, N.H., 1981. Properties of some protein fractions from bambara groundnut (Voandezeia subterranean) Thoura. J. Sci. Edn. Agric., 32: 44-50.
CrossRef - Zhang, D. and Y. Hamauzu, 2004. Phenolics, ascorbic acid, carotenoids and antioxidant activity of broccoli and their changes during conventional and microwave cooking. Food Chem., 88: 503-509.
CrossRefDirect Link








