Research Article
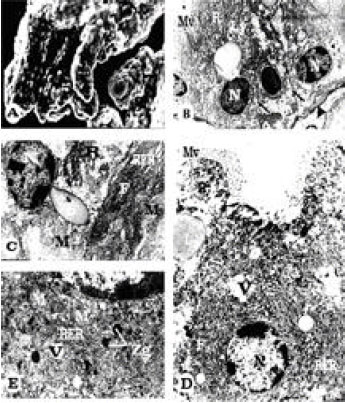
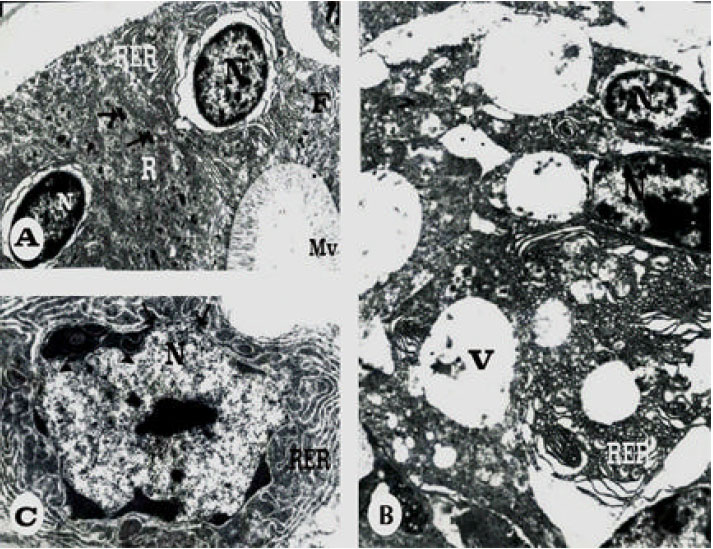
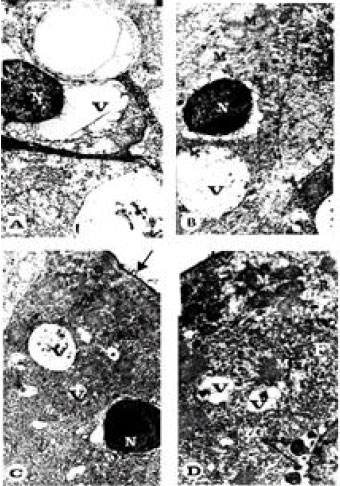
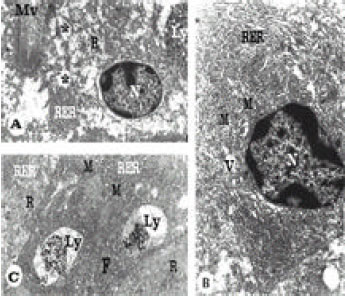
Ultrastructural Changes in Hepatopancreas of Palaemon serratus, Following Treatment with Petroleum Carcinogenic Compounds
Department of Zoology, Faculty of Science, Alexandria University, Alexandria, Egypt
Hassan E. Awad
Department of Oceanography, Faculty of Science, Alexandria University, Alexandria, Egypt
Ahmed M. Ibrahim
NIOF, Alexandria, Egypt
Nabiha A. Yousef
NIOF, Alexandria, Egypt