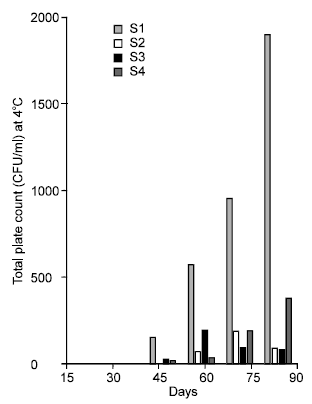
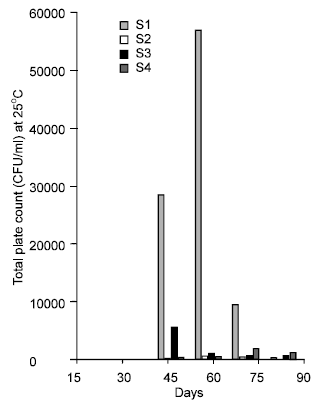
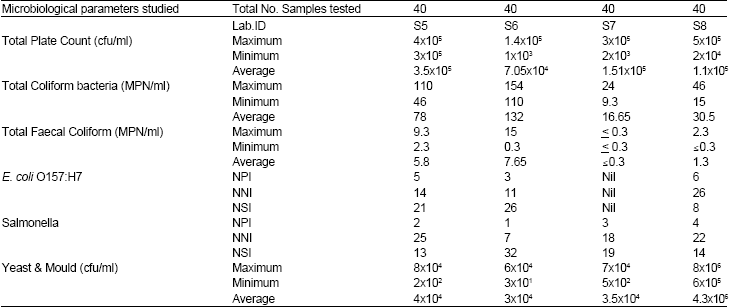
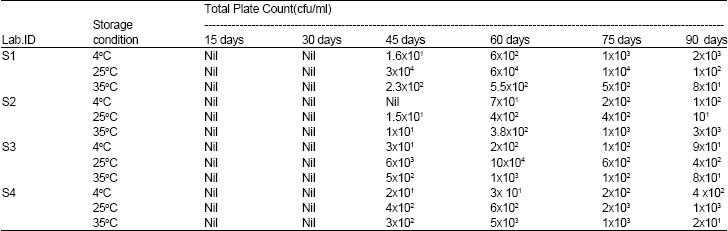
Research Article
Microbial Quality Assessment Study of Branded and Unbranded Milk Sold in Peshawar City, Pakistan
Food Technology Centre, PCSIR Laboratories Complex, Peshawar-25120, Pakistan
Javed Ali
Food Technology Centre, PCSIR Laboratories Complex, Peshawar-25120, Pakistan
Mohammad Waqas
Institute of Food Technology, NWFP Agriculture University, Peshawar-25120, Pakistan
Yasir Anwar
Food Technology Centre, PCSIR Laboratories Complex, Peshawar-25120, Pakistan
Javed Ullah
Institute of Food Technology, NWFP Agriculture University, Peshawar-25120, Pakistan