ABSTRACT
The germination responses of Hordeum ulgare seeds to saline stress caused by different salt types was studied. For this, 25 seeds of mentioned species were placed on filter paper in Petri dishes containing distilled water (control), 60, 120, 180, 240, 300, 360 and 420 mM. saline solution of NaCl, CaCl2 an KCl. The results indicated that saline levels effects were significant (P < 0.05) for seed germination percentage, seed germination velocity, mean time to germination, length of the stem and radicle and seed vigour. Seed germination decreased significantly by increasing salinity levels. Also, the results showed that H. vulgare is more tolerant than H. vulgare against salinity in germination stage.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjn.2009.63.68
URL: https://scialert.net/abstract/?doi=pjn.2009.63.68
INTRODUCTION
Soil salinity is a major factor limiting plant productivity, affecting about 95 million hectares world wide (Szabolcs, 1994). The UNEP (United Nations Environment Program) estimates that 20% of the agricultural and 50% of the cropland in the world is salt stressed (Flowers and Yeo, 1995). Salinity imposes serious environmental problems that affect grassland cover and the availability of animal feed in arid and semi-arid regions (El-Kharbotly et al., 2003). Salt stress unfavorably affected by plant growth and productivity during all developmental stages. For example Epstein et al. (1980) reported that salinity decreases seed germination, retards plant development and reduces crop yield. Shokohifard et al. (1989) reported that salt stress negatively affected seed germination; either osmotically through reduced water absorption or ionically through the accumulation of Na and Cl causing in imbalance in nutrient uptake and toxicity affect saline soils contain multiple types of soluble salt components, each of which has a different effect on the initial growth of plant (Redmann, 1974; Hardegree and Emmerich, 1990; Tobe et al., 2002) and the composition of soluble salts in saline soils differ greatly among locations (Tobe et al., 2002).
Although most of these reports are based on experiments with NaCl, it is hypothesized that other salts have similar effects on cellular function, but to different degrees, depending on the salt. Studies to examine salinity effects on the initial growth of plants have usually been carried out with individual salts (especially NaCl). Many authors have used NaCl solutions to study salinity tolerance in the germination of H.vulgare (Luque and Bingham 1981; Tsonev et al., 1998; Flowers and Hajidagheri 2001; Neslihan et al., 2002; Grant 2003; El-Tayeb 2005), but little information exists concerning the effect of other salts on the seed germination. However, there remains a question as to whether the results of experiments with individual salts simulate the actual initial growth behaviors of plants in saline soils. To clarify the responses of the seed germination and the initial growth of plants to salinity, examination of the effects of various salts is desirable. In the present study, we compared the effects of different types of salts on the seed germination of H.vulgare and H. vulgare. Because the main salt components of saline soils are Na+, Mg2+ and Ca2+ cations and cl- and SO42- anions (Shainberg, 1975), we investigated the effect of individual salts, NaCl, CaCl2 and KCl.
Materials and Methods
Study species: H.vulgare and H. vulgare belong to Graminae family. H.vulgare is widely grown in the arid and semi-arid regions of the Mediterranean for forage purposes and as grain crop (Al-Karaki, 2001). It is considered highly salt tolerant of the agriculturally important cereals and has been grown successfully in fields that irrigation has rendered unsuitable for other crops (Hopkins, 1995). Generally, it grown near soil surface where the salts accumulated and at this point of soil, the concentration of salt changes over time by continuous evapotranspiration gradually rising salt levels or rainfall leaching salts from the soil surface supplying water to seeds (Al-Karaki, 1997; Tobe et al., 2000a,b). H. vulgare is a perennial grass with large distribution in Iran. It can endure high-temperature, aridity and cooling (Moghimi, 2005).
Effects of salinity on germination: To evaluate salt tolerance during germination, 25 seeds of each species were placed on filter paper in 9 cm petri dishes and submerged in 5 ml of each solution. Solutions of the NaCl, CaCl2 and KCl were used at concentrations of 0 (control), 60, 120, 180, 240, 300, 360 and 420 mM. Experiments were performed in a completely randomized design with 4 replicates in the seed laboratory of Natural Resources Faculty of Tehran University.
Germination counts were made daily and were considered to have germinated when the radicle emerged. The water level was adjusted at 2-d intervals with distilled water to avoid changes in salinity due to evaporation. At the end of the germination period, the germination percentage, germination velocity, the mean time to germination and length of the stem and radicle under salinity were calculated. Germination percentage was calculated using the equation:
Also, germination velocity and the mean time to germination were calculated using the following equations:
Mean time to germination = (∑ni x di) /N, where n is the number of seeds germinated at day I , d the incubation period in days and N is the total number of seeds germinated in the treatment (Brenchley and Probert, 1998). Also, seed vigour was obtained using the following equation (Abdul-Baki and Anderson, 1973):
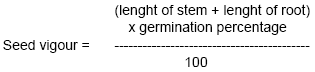 |
Statiscal analysis: A multrivariate ANOVA was used to evaluate the effects of salinity on seed germination. Data were analyzed using SPSS 11.5 for windows (SPSS Inc., 1999). When significant main effects existed, differences were tested by a multiple comparison Tukey test at 95% confidence. Germination data were arcsine transformed before statistical analysis to ensure homogeneity of variance.
Results
Effects of salinity on germination: Significant differences were obtained for three considered factors (species, salts and concentrations) and their interactions regarding seed germination (p < 0.05) (Table 1). The velocity of germination was also significant for three factors (species, concentrations and theirs interaction) and for the other three factors (salts, salts-species and salts-concentrations interaction) was not significant (Table 2). The mean time to germination was also significantly affected by all the factors (except to species) and interactions except that between species and concentrations (Table 3).
| Table 1: | Germination percentage of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
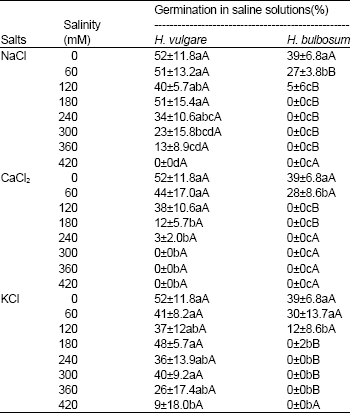 | |
| Values are mean±S.D. (n = 4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P < 0.05). | |
In the most of the treatments, H.vulgare showed higher germination percentage and germination velocity compared to H. bulbosum while its mean time to germination was less than H. vulgare. Generally, germination percentage and germination velocity were reduced by increased salt concentration. This condition was more intensity in H. vulgare than H.vulgare. For example, when the seeds were incubated with NaCl, concentrations higher than 120 mM, significantly reduced the germination of H. vulgare. This condition was similar to that of CaCl2 and KCl (Table 1). When seeds incubated with NaCl, in the most of the salinity concentrations, for both H.vulgare and H. vulgare, the mean time to germination was less than two other salts (CaCl2 and KCl). Generally, significant differences were obtained when comparing two similar salinity levels of different salts (for example, 60 mM solutions of NaCl and CaCl2 and etc).
Effects of salinity on length of the stem and radicle: The results showed that H.vulgare stem length was significantly bigger than that of H. vulgare by both salts
and salinity levels (Table 4). In both H.vulgare and H. vulgare, increase of each salt concentration caused decline of length of the stem. 60 mM treatment has shown the higher length of the stem following control (0 mM).
| Table 2: | Germination velocity of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
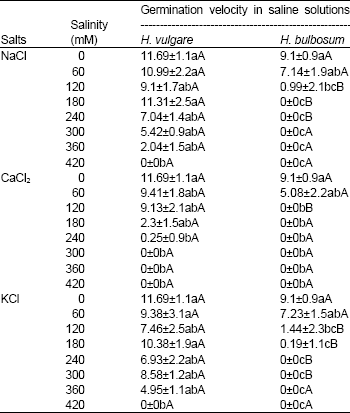 | |
| Values are mean±S.D. (n = 4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P < 0.05). | |
Although, in this treatment significant difference was not obtained among three salts. H. vulgare has shown length of the radicle more than H. vulgare and significant difference was obtained by all the treatments except for the control treatment (Table 5).
In both H. vulgare and H. bulbosum, length of the radicle was more in NaCl than CaCl2 and KCl by all the salinity levels except to the control treatment (Table 5).
Generally, significant difference was obtained for the salts. Similar to length of the stem, length of the radicle also reduced by increase salinity levels in both species H. vulgare and H. bulbosum and three salts (Table 5). Length of radicle was significantly different comparing with the control and other treatments in both species and three salts except to of NaCl salt in H. bulbosum. Length of the stem showed similar behavior for H. vulgare, but in H. bulbosum, significant difference was not obtained among control treatment with 60 mM in three salts.
NaCl, CaCl2 and KCl concentrations of 120 mM and higher salinity levels sharply reduced the length of the stem of H. vulgare, but this condition (Sharp reduction) was obtained at a concentration of 60 mM and higher levels for H. vulgare.
| Table 3: | Mean time to germination of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
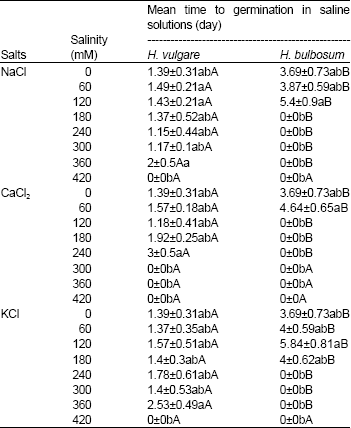 | |
| Values are mean±S.D. (n = 4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P< 0.05). | |
In spite of the length of the stem of H. vulgare, reduction of length of the radicle wasn’t very clear (except to CaCl2). On the other hand, reduction in tength of the radicle due to increase of the salinity levels was gradually and nonclear. Adversely, this difference was clear. For this reason, significant and large difference was obtained in comparing control treatment with other treatments.
Seed vigor: The principal object of a seed vigor test is to differentiate a range of quality levels, for example, high, medium and low vigor seeds. The results obtained through germination test by way of evaluating likely seed performance (plant stand) under a wide range of field conditions (Rattan, 1998). As it has been represented in equation of seed vigor, seed vigor is obtained by germination percentage and length of stem and radicle, therefore the results of seed vigor is largely similar to them. However, as it is shown in Table 6, a significant difference is observed among species, salts and salinity levels. The seed vigor of H. vulgare was higher than H. bulbsum. In all of salts in different levels from 0-420 mM, the general trend is associated with seed vigor decrease. In H. vulgare and H. bulbosum, among three salts, it seems that CaCl2 has the largest effect on seed vigor, so that no seed vigor is considered offer 240 and 120 mM, respectively.
| Table 4: | Stem length of seeds of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
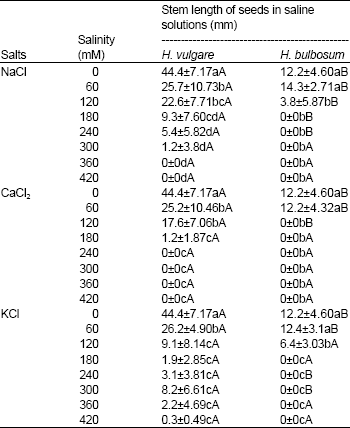 | |
| Values are mean±S.D. (n=4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P < 0.05). | |
DISCUSSION
As showed in results, H. vulgare was more salt-tolerant than H. bulbosum. For H. vulgare, although maximum germination was obtained under non-saline conditions (control treatment), its seeds had the ability to germinate at higher levels of salinity in NaCl and KCl, especially, but in H. bulbosum, intent reduction in germination was obtained at 180mM level and above. As referred in results, seed germination reduced by increasing salinity levels, except at salinity levels of KCl related to H. Vulgare.
Reduction in germination by an increase of salinity levels has been described by numerous authors (Othman et al., 2006; El-Tayeb, 2005; Breen et al., 1997; Ungar, 1982).
Considerable variations were observed between two species especially at higher salinity levels in response to salinity for germination percentage. These results were in agreement with Basalah (1991) who found that high levels of salinity can significantly inhibit seed germination.
| Table 5: | Radicle length of seeds of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
 | |
| Values are mean ± S.D. (n=4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P < 0.05). | |
Further, Waisel (1972) found that increasing salinity concentration in germination often cause osmotic and/or specific toxicity which may reduce or retard germination percentage. Significant difference was obtained for salts regarding seed germination. The behavior (reduction or increase in seed germination by increase of salinity levels) of three salts which studied in this research, was different and partially intricated. As the results demonstrate, this significant difference was at higher levels, especially, while it wasn’t significant for under levels such 0 mM (control treatment), 60 and 120 mM. In our research, at above levels maximum germination was obtained for KCl, H. vulgare, especially, thus we can tell that salt tolerance of seed to this salt is partially more than two other salts. It could be concluded that k+ ion accumulation in the cytosolic solutes reduce more negative effect of cl- than Na+ ion. NaCl affects the permeability of the plasma membrane and increases influx of external ions and efflux of cytosolic solutes (Cramer et al., 1985; Kent and Lauchli, 1985; Allen et al., 1995) in plant cells. In addition, NaCl causes hardening of the cell wall (Neumann, 1993; Neumann et al., 1994; Nabil and Coudret, 1995) and a decrease in water conductance of the plasma membrance (Azaizeh et al., 1992; Cramer, 1992). These effects of NaCl on cellular function are alleviated by the addition of Ca2+ to the external medium (Cramer et al., 1985; Kent and Lauchli, 1985; Azaizeh et al., 1992; Cramer, 1992; Allen et al., 1995).
| Table 6: | Seed vigor of Hordeum vulgare and Hordeum bulbosum seeds in saline solutions of NaCl, CaCl2 and KCl |
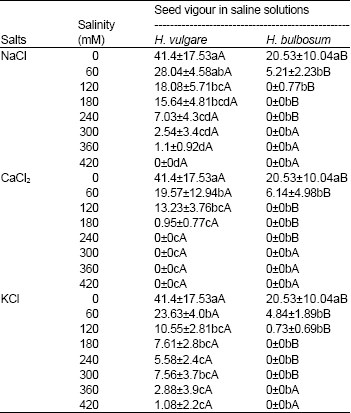 | |
| Values are mean±S.D. (n=4). Means within a column that have a different small letter are significantly different from each other, and means within a row that have different capital letter are significantly different from each other (Tukey test; P< 0.05). | |
These effects of salts on the function of the cell membranes and the walls may affect the water potential of the cytosol and cellular extensibility and thus, may affect seed germination, while we found an unexpected result in our research and that is reduction in germination by CaCl2 in both H.vulgare and H. bulbosum. Probably, we can conclude that large amount of Ca2+ caused an negative effect on germination. Generally, this differential behavior of seeds according to the salt type is also presumably due to the fact that the same concentration of salt generates different osmotic potentials and the osmotic effect may well have a greater influence on germination than specification toxicity, as has been suggested by several authors in other plants such much of halophytes (Unger, 1996; Pujol et al., 2000).
In general, based on the obtained results it seems that H. vulgare is more tolerant than H. bulbosum against different salts. Therefore, between two mentioned species, using H. vulgare seeds in range reclamation and restoration projects could be lead to suitable results in arid and semi arid areas, where salinity affects vegetation development plans strongly.
REFERENCES
- Abdul-Baki, A.A. and J.D. Anderson, 1973. Vigor determination in soybean seed by multiple criteria. Crop Sci., 13: 630-633.
CrossRefDirect Link - Allen, G.J., R.G.W. Jones and R.A. Leigh, 1995. Sodium transport measured in plasma membrane vesicles isolated from wheat genotypes with differing K+/Na+ discrimination traits. Plant Cell Environ., 18: 105-115.
CrossRef - Al-Karaki, G.N., 2001. Germination, sodium and potassium concentrations of barley seeds as influenced by salinity. J. Plant Nutr., 24: 511-522.
CrossRefDirect Link - Al-Karaki, G.N., 1997. Barley response to salt stress at varied levels of phosphorous. J. Plant Nutr., 20: 1635-1643.
CrossRefDirect Link - Breen, C.M., C. Everson and K. Rogers, 1997. Ecological studies on Sporobolus virginicus (L.) Kunth with particular reference to salinity and inundation. Hydrobiologia, 54: 135-140.
CrossRef - Brenchely, J.L. and R.J. Probert, 1998. Seed germination responses to some environmental factors in sea grass Zostera capricorni from Eastern Australia. Aquat. Bot., 62: 177-188.
CrossRef - Cramer, G.R., 1992. Kinetics of maize leaf elongation. II. Responses of Na-excluding cultivar and a Na-including cultivar to varying Na/Ca salinities. J. Exp. Bot., 43: 857-864.
Direct Link - El-Kharbotly, A., O. Mahgoub, A. Al-Subhi and A. Al-Halhali, 2003. Indigenous grass species with potential for maintaining rangeland and livestock feeding in Oman. Agric. Ecosyst. Environ., 95: 623-627.
Direct Link - El-Tayeb, M.A., 2005. Response of barley grains to the interactive e.ect of salinity and salicylic acid. Plant Growth Regul., 45: 215-224.
CrossRefDirect Link - Epstein, E., J.D. Norlyn, D.W. Rush, R.W. Kingsbury, D.B. Kelley, G.A. Cunningham and A.F. Wrona, 1980. Saline culture of crops: A genetic approach. Science, 210: 399-404.
CrossRefDirect Link - Flowers, T.J. and A.R. Yeo, 1995. Breeding for salinity resistance in crop plants: Where next? Aust. J. Plant Physiol., 22: 875-884.
CrossRefDirect Link - Hardegree, S.P. and W.E. Emmerich, 1990. Partitioning water potential and specific salt effects on seed germination of four grasses. Ann. Bot., 66: 587-595.
CrossRefDirect Link - Kent, L.M. and A. Lauchli, 1985. Germination and seedling growth of cotton: Salinity-calcium interactions. Plant Cell Environ., 8: 155-159.
CrossRefDirect Link - Nabil, M. and A. Coudret, 1995. Effects of sodium chloride on growth, tissue elasticity and solute adjustment in two Acacia nilotica subspecies. Physiol. Plant., 93: 217-224.
CrossRef - Neumann, P.M., H. Azaizeh and D. Leon, 1994. Hardening of root cell walls: A growth inhibitory response to salinity stress. Plant Cell Environ., 16: 303-309.
CrossRefDirect Link - Othman, Y., G. Al-Karaki, A.R. Al-Tawaha and A. Al-Horani, 2006. Variation in germination and ion uptake in barley genotypes under salinity conditions. World J. Agric. Sci., 2: 11-15.
Direct Link - Shokohifard, G., K.H. Sakagam, R. Hamada and S. Matsumoto, 1989. Effect of amending materials on growth of radish plant in salinized soil. J. Plant Nutr., 12: 1195-1214.
CrossRef - Tobe, K., X. Li and K. Omesa, 2000. Seed germination and radicle growth of a halophyte, Kalidium capsicum (Chenopodiaceae). Ann. Bot., 85: 391-396.
CrossRefDirect Link - Ungar, I.A., 1996. Effect of salinity on seed germination, growth and ion accumulation of Atriplex patula (Chenopodiaceae). Am. J. Bot., 83: 604-607.
Direct Link








