Research Article
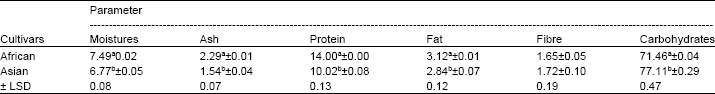
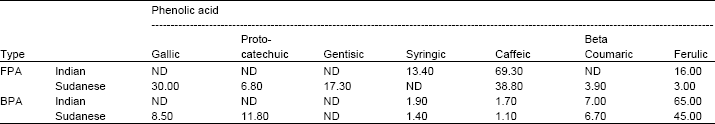
Characterization of Tannin and Study of in vitro Protein Digestibility and Mineral Profile of Sudanese and Indian Sorghum Cultivars
Department of Food Science and Technology, Faculty of Agriculture, University of AL Zaim AL-Azhari, Khartoum, Sudan
G. Muralikrishna
Department of Biochemistry and Nutrition, CFTRI, Mysore 570 013, Karnataka, India
A.H. EL Tinay
Department of Food Science and Technology, Faculty of Agriculture, University of Khartoum, Sudan
A.I. Mustafa
Department of Food Science and Technology, Faculty of Agriculture, University of Khartoum, Sudan