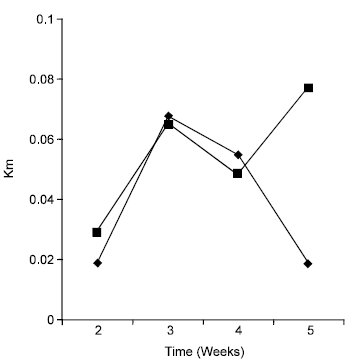
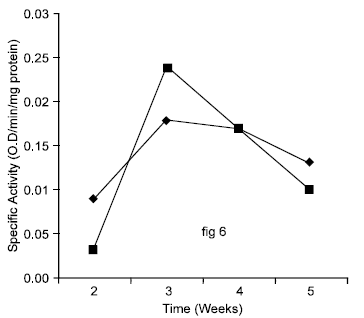
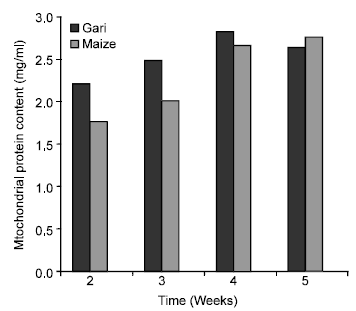
Research Article
Effect of Gari Diet on Marker Enzymes of Mice Liver Mitochondria
Department of Biotechnology,
O. Obidua
Department of Biochemistry, University of Nigeria, Nsukka, Nigeria
I.G. Kalu
Department of Biotechnology,
I.N. Nwachukwu
Department of Microbiology, Federal University of Technology, P.M. B. 1526, Owerri, Nigeria