Research Article
Effect of Carrot Extracts on Pseudomonas aeruginosa
Department of Veterinary Public Health, Veterinary Medicine College, University of Baghdad, Baghdad, Iraq
Pseudomonas aeruginosa is one of the most zoonotic and pathogenic bacteria which infect man and animals and others (Ryan and Ray, 2004; Iglewski, 1996), Causing many infections in humans particularly in immunoompriminized individuals. The bacteria infect the respiratory system causing respiratory problems as pneumonia, cystic fibrosis; the bacteria also infect the urinary system, infection of burns and wounds.
P. aeruginosa is widely distributed and considered as one of the nosocomial infections; the bacteria can resist many antibiotics due to it's genetic factor (Cornialis, 2008; Todar`s, 2008).
The plants and their extracts were used since thousands years ago for many purposes of food and as natural preservatives from oxidation and decay; some of plants can be used as antimicrobial in food with different spectrum as natural drugs preparations (Hammer et al., 1999; Brul and Coote, 1999; Cowan, 1999; Lima et al., 2005). Carrot is one of the plants; the scientific name is daucus carota subsp. Sativus. This plant is natural foods from the genus apiaceae (Ahmed et al., 2005), this plant is formed as plant root, Horney shape with orange, red or white with crispy shape in a fresh state, the natural habitat in Europe and West-South Asian countries, carrot is rich in carbohydrate, carotene, vitamins, minerals and others and has a white flowers appear on the surface of soil, this plant used in many foods as fresh or boiled especially in the preparation of broths with onion juices and a juices in which the carrot juices considered as one of the healthy drink, which is beneficial for humans particularly in fresh state as alone or mixed with fruits or milk. Carrot is rich in Vitamin A which is useful in vision health and as antioxidants which preserves food from decay and keeps the health and immunity of humans from the risk of oxidative contaminants (Carrot, 2008). The other medical uses for the roots of carrots and their extracts can be used for the treatment of disturbances and diseases of gastrointestinal tract for constipation, carminatives and as antihelmintics, also carrots and its extracts can be used as antiseptic for wounds and activator for gastrointestinal tract and anti-inflammatory especially in tonsillitis and as hepato-protective and in treatment of jaundice and dermatitis (Ahmed et al., 2005; Carrot, 2008), also it was found that the carrots and its extracts has effect against the microbes as bacteria and fungi (Hammer et al., 1999; Ahmed et al., 2005; Lima et al., 2005; Carrot, 2008).
Recently an oily alcohol extract obtained from carrot and from Read-ginser plant which have a strong effect against tumors especially the tumor of urinary tract and skin tumors (Ahmed et al., 2005; Carrot, 2008). Due to constituents of many active ingredients especially sesquterpenes daicanes with different types, carotol, sitasterol-juicosidies, fatty oils, flavonoids, polyacetyenes, β-carotenes, furocoumarins and other compounds (Ahmed et al., 2005).
Culture isolate: Pseudomonas aeruginosa which was obtained from the unit of zoonosis, College of Veterinary Medicine, University of Baghdad.
Carrots: The carrot was obtained from the local Markets in Baghdad and the extracts were prepared in the unit of zoonosis.
Culture media: Trypticase Soya yeast extract with 0.6% of the yeast was obtained from Oxoid Company.
Blood agar base: From (Oxoid Company) in which 7 mL of blood sheep was added to prepare blood Agar.
Nutrient broth: (From Oxoid).
Mueller-hinton agar: (From Biolife company), in which was used for the sensitivity of the bacteria against antibiotics.
Gelatinase: (From Difco company) used to confirm that the bacteria was liquefactant.
Solutions
Gram stain: (From Crescent Company).
PBS: Prepared in the laboratory.
Oxidase indicator (from Fluka Company) which was used to differentiate between Pseudomonas aeruginosa and interobacteriaceae.
Absolute alcohol-ethanol: (from Fluka Company).
Sterile-cotton swabs: (from Himedia Company).
Antimicrobial susceptibility discs: (from Bioanalyse). Which include 10 μg Ampicillin, 30 μg Tetracycline, 5 μg Trimethprim.
The confirmations of Pseudomonas aeruginosa was done by culturing on Trypticase Soya yeast extract agar and incubation at 37oC for 24 h and then biochemical tests as oxidase, Gelatinase, motility test, hemolysis on blood agar and staining with gram stain.
Carrot extracts: Prepared in the laboratory of Zoonosis unit by using cold methods extract according to AOAC (1980).
The standard culture method: This method was done according to (National Committee for Clinical Laboratory Studies, 1990). By selecting five pure colonies from Pseudomonas aeruginosa and transferred to a test tube contain 5 mL of TSE-YB broth and incubated at 37oC for 24 h, after that the turbidity of the cultured broth compared with McFarland standard (tube no.5) to obtain a culture with density of 105-106 cfu of the bacteria. The number of bacteria was confirmed by using (Miles et al., 1993). Method the culture of the plates and incubation at 37oC for 24 h and counted the number of bacteria 105-106/cfu.
The sensitivity test: The method of Bauer et al. (1966) was used for the purpose of sensitivity of bacteria for the antibiotics by the wells diffusion and discs techniques:
The well-technique: After the preparation of the cultures media TSE-YB in Petri plates were heavily inoculated with a culture broth of the bacteria by taking swab from the culture broth and spreaded on the surface of the agar medium (this method of taking swabs) repeated four times, for each plate in order to obtain thick culture after that 5 wells were made in each plate by using coric
pierans and then drops of each extract (alcoholic and watery extracts of carrots) nearly 0.5 mL for each well in plates, then left to dry, after that invert the plates and incubated at 37° C for 24 h.
The use of filter papers impregnated technique: (small and large) which impregnated with each of alcoholic and watery extracts of carrots with the testing of sensitivity of the Ampicillin, Tetracycline and Trimethprim. This method was done by impregnated the Standard small filter paper with each of extracts for 15 min and transferred on the surface of the solid medium which was inoculated with Pseudomonas aeruginosa, one filter paper used for each plate and for each extract with a plates containing the antibiotics for the purpose of comparison.
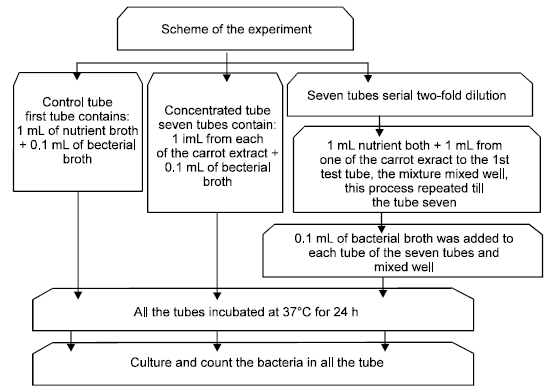
Estimation of Minimal Inhibitory Concentration (MIC): The method of dilution in broth culture medium according to (National Committee for Clinical Laboratory Studies, 1990) was used to estimate the Minimal Inhibitory Concentration (MIC). This method was used by two-fold dilution and ten-fold dilution. The two-fold dilution for extracts broth and as follows:
The control tube contains 1 mL of the sterile nutrient broth + 0.1 mL of Pseudomonas aeruginosa broth..
The concentrated tube (final tube) contains 1ml from one of the concentrated extracts + 0.1 mL from standard broth of Pseudomonas aeruginosa.
Seven tubes contain serial two-fold dilution + 1 mL of the sterile nutrient broth was added for each test tube + 1 mL from one of the concentrated extracts to the first test tube only of the seven tubes then the mixture mixed well by rotomixer for 5 min, after that transfer 1 mL from the mixture to the second test tube, this method of transferring repeated till the seventh test tube after that a standard and limited amounts (0.1 mL) from the inoculum's of bacteria was added to each tube till the Seventh tube. After that all the tubes were incubated at 37oC for 24 h, after that culturing and count of the bacteria were performed by using TSE-YA medium and compared with control and concentrated tubes for the effect of the extracts.
Ten-fold dilution method for the carrot extracts by the use of Standard nutrient broth and this method performed as the previous method, but the 7th tubes contained 9 mL from the Standard nutrient broth and the addition of 1 mL from one of the carrot extracts to the first test tube and mixed well, after that transfer 1 mL from the mixture to the second test tube, this process repeated in all the seventh test tubes, after that 0.1 mL from the bacterial broth transferred to each of the seventh test tubes and mixed well and all tubes incubated at 37oC for 24 h, after that the number of bacteria was counted in each test tube as previously mentioned.
The results of sensitivity tests by filter papers (small and large) and by well method for the alcoholic and watery extracts of carrots showed inhibition zones which were more clear and large in alcoholic extract than the watery extract. This gave indication that the carrot extracts have inhibitory effect on the growth of Pseudomonas aeruginosa in vitro as shown in Table 1. Whereas, this bacteria showed resistance to the antibiotic used in this experiment as shown in Table 2. This explained that there was a natural and efficient substance which is capable to inhibit the growth of bacteria in which can resist the antibiotics (Hammer et al., 1999; Dorman and Dean, 2000; Lee et al., 2004; Ahmed et al., 2005).
The effect of carrot extracts on the inhibition of growth of Pseudomonas aeruginosa particularly the alcoholic extract related to many factors as plant genus, the period of ripening, type of the soil, climatic factors and the chemical structure of the extracts also to the presence of the active ingredient in the extract and to the type of microorganism affected by the plant extracts (Janssen et al., 1987; Dorman and Dean, 2000; Lee et al., 2004).
The results of Minimum Inhibitory Concentration (MIC) on the growth of Pseudomonas aeruginosa for alcoholic and watery extracts, showed a decrease in the growth about 0.2 log. Ten 10 For the concentration 25 and 50% and 0.4 log. In a concentration 100%.
It was found that the alcoholic extract has more effect on the growth of bacteria which explain the efficiency of carrot to reduce the contamination by Pseudomonas aeruginosa (Ahmed et al., 2005).
The effect of the carrot and its extracts can be explain due to many interacting factors as the presence of zerpinene and other active ingredient which have chemical nature capable to impair the activity or growth of the bacteria due to the effect on the internal contents of the bacteria which cause the inhibition of growth or killing of the pathogenic bacteria (Janssen et al., 1987; Lee et al., 2004) as shown in Table 3 and 4.
| Table 1: | The sensitivity tests by the filter papers and well diffusion methods |
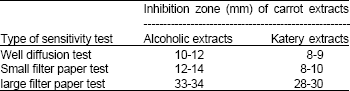 | |
| Table 2: | The sensitivity test of Pseudomonas aeruginosa against the antibiotics |
 | |
| R: resistant. I: Intermediate | |
| Table 3: | The effect of the lowest carrot watery extract on the inhibition Pseudomonas aeruginosa |
 | |
 |
| Table 4: | The effect of the lowest carrot alcoholic extract on the inhibition Pseudomonas aeruginosa |
 | |