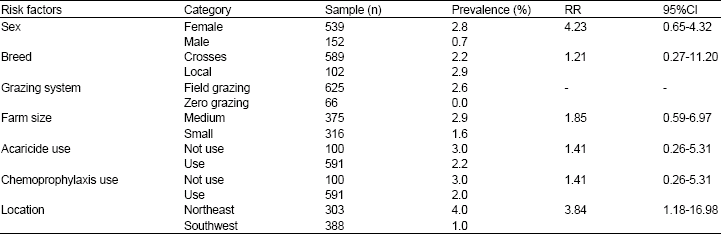
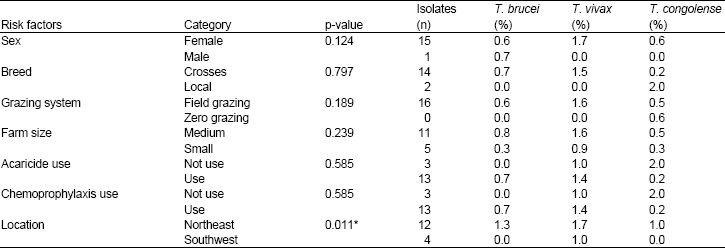
Research Article
Prevalence of Bovine Trypanosomosis in Morogoro, Tanzania
Department of Veterinary Medicine and Public Health, Faculty of Veterinary Medicine, Sokoine University of Agriculture, P.O, Box 3021, Morogoro, Tanzania
D.M. Kambarage
Department of Veterinary Medicine and Public Health, Faculty of Veterinary Medicine, Sokoine University of Agriculture, P.O, Box 3021, Morogoro, Tanzania