ABSTRACT
Effects of aqueous extract of Ziziphus mauritiana (Zm) leaf on serum and liver cholesterol and triacylglycerol were studied in chronic alcohol administered rats. Pretreatment co-administration and post treatment protocols were employed. Rats fed alcohol only for 6 weeks had significantly (p<0.05) elevated levels of both serum and liver cholesterol and triglyceride. Pretreatment of rats with 400 mg/kg bw of aqueous extract of Zm leaf prior to alcohol administration significantly (p<0.05) inhibited both levels of serum and liver cholesterol and triglyceride. Co-administration of alcohol with 400 mg/kg bw aqueous extract of Zm leaf significantly (p<0.05) inhibited serum triglyceride level only. Post treatment of alcohol fed rats with 400 mg/kg aqueous extract of Zm for 2 weeks significantly (p<0.05) reduced the levels of both serum and liver cholesterol and triglyceride. Aqueous extract of Zm leaf can be used for the prevention and treatment of fatty liver, atherosclerosis and other diseases associated with high levels of cholesterol and triglyceride. Pretreatment was found to confer more protection than co-treatment, hence pretreatment should be preferred.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjn.2009.1884.1888
URL: https://scialert.net/abstract/?doi=pjn.2009.1884.1888
INTRODUCTION
The pathogenesis of alcoholic fatty liver and alcoholic hyperlipidemia has been known for a long time to be due mainly to a combination of decreased fatty acid oxidation in mitochondria and to increased glycerolipid synthesis (Baraona and Lieber, 1998). Alcohol intake is only second to diabetes mellitus as a cause of hyperlipidemia which is relevant to the problem of atherosclerosis and heart disease in the drinking population (Feinman and Lieber, 1992). In the general population, elevations in LDL cholesterol are correlated with increase risk of coronary artery disease while hypertriglyceridemia is seen mostly in patients with fatty liver. Elevated serum triglycerides have been identified as an independent risk factor for cardiovascular disease. In view of the effect of ethanol on lipid metabolism especially on cholesterol and triglyceride with its consequent adverse effects, we decided to study the effect of aqueous extract of Ziziphus mauritiana leaf on cholesterol and triglyceride levels in serum and liver of rats administered alcohol. Three protocols, pretreatment, co-administration and post-treatment were employed.
Ziziphus mauritiana Lam belongs to the family Rhamnaceae. It is commonly known as ‘magarya’ in Hausa (Nigeria) and is used both as a source of food and medicine. The plant is a vigorous grower and has a rapidly developing taproot. It may be a bush shrub four to six ft (1.2-1.8m) high, or a tree 10 to 30 or even 40 ft (3-9 or 12 m) tall; erect or wide-spreading, with gracefully drooping branches and downy, zigzag branch-lets, thorn-less or set with short, sharp straight or hooked spines. It may be evergreen or leafless for several weeks in hot summers. The leaves are alternate, ovate or oblong-elliptic, one to two and a half inches (2.5-6.25 cm) long, three quarter to one and a half inches (2-4 cm) wide. On the upper surface, they are very glossy, dark-green, with three conspicuous, depressed, longitudinal veins and there are very fine serrated margins (Morton, 1987; Michel, 2002).
The leaves are applied as poultices and are helpful in liver troubles, asthma and fever. The bitter, astringent bark decoction is taken to halt diarrhea and dysentery and relieve gingivitis. The back paste is applied on sores. The root is used as a purgative. A root decoction is given as a febrifuge and the powdered root is dusted on wounds. Juice of the root bark is said to alleviate gout and rheumatism. An infusion of the flowers serves as an eye lotion (Morton, 1987; Michel, 2002). Dahiru et al. (2004) reported the antisalmonella activities of the methanol and aqueous extracts of Ziziphus mauritiana leaf. Protective effect against carbon tetrachloride induced liver injury (Dahiru et al., 2005), protective effect against alcohol induced liver damage (Dahiru and Obidoa, 2007).
MATERIALS AND METHODS
Plant material: The leaf of Ziziphus mauritiana was collected along Yola-Mubi road Adamawa State. The leaves were allowed to dry under shade before being ground into fine powder using mortar and piston.
| Table 1: | Effect of pre-treatment with Ziziphus mauritiana aqueous leaf extract on serum and liver cholesterol and triglyceride in chronic alcohol-induced liver damage |
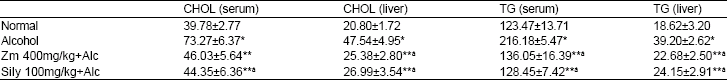 | |
Results are Mean±S.D (n = 5). *significantly higher than normal group (p<0.05). **significantly lower than Group administered alcohol only. αsignificantly lower than group pre-treated with 200 mg/kg aqueous extract of Ziziphus mauritiana (p<0.05) | |
The powder was then sieved using Endecott® test sieve (0.3 mm mesh diameter) [Endecott Ltd. London]. The sieved powdered material was kept in a dark plastic container prior to use.
Animals: Male Wistar rats weighing between 100-120g were used for the studies. The animals were purchased from the Veterinary Research Institute Vom, Plateau State, Nigeria. They were kept under standard conditions of 12/12h dark/light cycle and were fed with standard growers feed (Vitals Ltd Jos, Nigeria) and water ad libitum.
Treatment
Protocol A (pretreatment): Twenty four male rats were distributed into 4 groups (6 rats/cage). Group I was the normal control and was given in addition to normal diet an isocaloric solution (1ml of 30% glucose/100g bw), group II experimental control (1 ml/100g, 40% v/v alcohol solution), groups III received 400mg/kg bw extract 30min prior to alcohol administration daily, while group IV received 100mg/kg (bw) of silymarin (standard drug) 30 min before alcohol administration. Both alcohol and extracts were administered for 6 weeks (42 days) by gastric intubations. Alcohol was given at 40% ethanol [v/v, 1ml/100g bw p.o].
Protocol B (co-treatment): Twenty four male rats were distributed into 4 cages (6 rats/cage) and labeled groups 1-4. Group I animals served as the normal control and received in addition to normal diet isocaloric solution (1ml of 30% glucose/100g bw), group II was the experimental control (alcohol only), group III was co-administered alcohol and 400 mg/kg bw of the extract while group IV was co-administered alcohol and 100 mg/kg bw silymarin in alcoholic solution daily. Both alcohol and extract were administered for 6weeks (42 days).
Protocol C (post-treatment): Twenty four male rats were distributed into 4 cages (6 rats/cage) labeled groups I-IV. Group I was the normal control and received in addition to normal diet an isocaloric solution (1 mL of 30% glucose/100g), group II was the experimental control (alcohol only), groups III and IV were administered alcohol for 6 weeks followed by treatment with 400 mg/kg aqueous extract of Ziziphus mauritiana leaf and silymarin 100 mg/kg bw respectively for two weeks.
Preparation of plant extract: A 100g of the air-dried ground leaf powder was steeped in 600 mL of hot distilled water and allowed to stand for 30 min. The extract was decanted into a clean measuring cylinder and the residue rinsed exhaustively with known volumes of hot water, the extract and the rinse water were pooled and filtered through Whatman No 4 filter paper under suction pressure. Thereafter, the filtrate was freeze-dried and weighed.
Biochemical estimations: At the end of the experimental period, the animals were sacrificed after ether anesthesia and blood collected without the use of anti-coagulant for serum preparation. The blood collected was allowed to stand for 10 min before being centrifuged at 2,000 rpm for 10 min after which the serum was collected using a rubber micropipette and used for the analysis. Cholesterol was determined after enzymatic hydrolysis and oxidation based on the method of Abell et al. (1952). Samples of liver tissues for analysis were homogenized in a chloroform-methanol mixture (1:1). Triglyceride was determined after enzymatic hydrolysis with lipases as described (Tietz, 1990). Samples of liver tissue for analysis were homogenized in a chloroform-methanol mixture (2:1).
RESULTS
The effects of pretreatment with aqueous extract of Ziziphus mauritiana leaf on serum and liver cholesterol and triglyceride levels in alcohol fed rats are presented in Table 1. Administration of alcohol alone to rats significantly (p<0.05) increased both levels of serum and liver cholesterol compared to normal rats. Pretreatment of rats with 400 mg/kg bw of Ziziphus mauritiana leaf extract significantly (p<0.05) decreased the raised levels of serum and liver cholesterol when compared to the group administered alcohol only. Pretreatment with the standard, drug, silymarin also significantly (p<0.05) decreased the level of serum and liver cholesterol when compared to the group II, but without any significant change when compared to group pretreated with 400 mg/kg bw of aqueous extract of Ziziphus mauritiana leaf. Pretreatment of rats with 400 mg/kg bw aqueous extract of Ziziphus mauritiana leaf significantly (p<0.05) decreased the levels of both serum and liver triglyceride compared to the group given alcohol only.
| Table 2: | Effect of co-treatment of alcohol with aqueous extract of Ziziphus mauritiana leaf on serum and liver cholesterol and triacylgycerol |
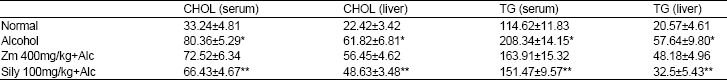 | |
Results are Mean±S.D (n = 5). *significantly higher than normal group (p<0.05). **significantly lower than group administered alcohol only (p<0.05) | |
| Table 3: | Effect of post-treatment with aqueous extract of Ziziphus mauritiana leaf on serum and liver cholesterol and triglyceride in chronic alcohol-induced liver damage |
 | |
Results are Mean±S.D (n = 5). *significantly higher than normal group (p<0.05). **significantly lower than alcohol untreated group (p<0.05). ásignificantly lower than group post-treated with 400 mg aqueous extract of Ziziphus mauritiana leaf (p<0.05) | |
Significant decrease (p<0.05) in the level of serum and liver triglyceride were observed in the group pretreated with silymarin compared to group to group administered alcohol only.
Table 2 show the results of co-treatment of alcohol with aqueous extract of Ziziphus mauritiana leaf on serum and liver cholesterol and triglyceride levels respectively. Administration of alcohol only to rats significantly (p<0.05) increased the levels of serum and liver cholesterol compared to the level in normal rats. Co-administration of alcohol with 400 mg/kg bw of aqueous extract of Ziziphus mauritiana leaf to rats did not show any significant change in the serum and liver cholesterol levels compared to the levels in the group fed alcohol only. However, co-treatment of alcohol with silymarin to rats (GP III) produced a significant (p<0.05) decrease in the levels of both serum and liver cholesterol compared to the group administered alcohol only. Levels of triglyceride in both serum and liver were significantly (p<0.05) elevated in rats given alcohol only compared to the levels in normal rats. Co-treatment of rats with ethanol and aqueous extract of Ziziphus mauritiana leaf significantly (p<0.05) reduced the increased level of serum triglyceride without any significant change in liver triglyceride level compared to rats administered alcohol only. Rats co-administered alcohol with silymarin also showed a significant (p<0.05) decrease in both serum and liver triglyceride levels compared to the group given alcohol only. A significantly (p<0.05) decreased level of serum triglyceride without any significant change in the level of liver triglyceride was observed in the group co-administered with alcohol and silymarin compared to the group co-administered alcohol with 400 mg/kg bw of aqueous extract of Ziziphus mauritiana leaf.
The effects of post-treatment with aqueous extract of Ziziphus mauritiana leaf on serum and liver cholesterol and triglyceride levels were analyzed and the results are presented in Table 3. Rats post-treated with distilled water had significantly (p<0.05) increased levels of both serum and liver cholesterol compared to the normal rats. Post-treatment of rats with 400 mg/kg bw aqueous extract of Ziziphus mauritiana leaf significantly (p<0.05) decreased the raised levels of serum and liver cholesterol compared to rats post-treated with distilled water. Post-treatment with the standard drug, silymarin significantly (p<0.05) decreased the observed increase in serum and liver cholesterol due to alcohol toxicity when compared with the group post-treated with 400 mg/kg bw of aqueous extract of Ziziphus mauritiana leaf. Group given alcohol only and later post-treated with distilled water had significantly (p<0.05) elevated levels of both serum and liver triglycerides compared to the normal group. Post-treatment of rats with 400 mg/kg bw of aqueous extract of Ziziphus mauritiana leaf significantly (p<0.05) lowered the raised levels of both serum and liver triglycerides when compared with rats administered alcohol but post-treated with distilled water only. Post-treatment with the standard drug, silymarin also significantly (p<0.05) decreased the level of both serum and liver triglycerides compared to the group post-treated with distilled water and without significant change when compared with group post-treated with 400mg/kg aqueous extract of Ziziphus mauritiana.
DISCUSSION
Chronic ingestion of alcohol in rats significantly increased the levels of cholesterol and triglyceride in both serum and liver. Marked alterations of lipid metabolism were reported in animals fed alcohol (Day et al., 1993). The development of fatty liver and hyperlipidaemia frequently occurs in chronic alcoholics mainly because ethanol becomes a preferred fuel for the liver and displaces fat as a source of energy, which results in fat accumulation. Furthermore, the redox state secondary to alcohol oxidation is altered coupling lipogenesis through increasing α-glycerophosphate and acylglycerols. The depressed oxidative capacity of the mitochondria caused by chronic alcohol ingestion also contributes to fatty liver.
During alcohol oxidation mediated by alcohol dehydrogenase (ADH), hydrogen is transferred from the substrate to the cofactor Nicotinamide Adenine Dinucleotide (NAD), converting it to its reduced state NADH. The excess of reduced equivalent, mainly NADH produces a change in the lactate/pyruvate ratio. This redox imbalance is responsible for a series of metabolic alterations which favour liver damage. AcetylCoA carboxylase (ACC) is an enzyme that catalyses the first step in fatty acid biosynthesis and is the rate limiting enzyme in lipogenesis. Moreover, Malic Enzyme (ME), glucose-6-phosphate dehydrogenase (G6PDH) and 6-phosphogluconic acid dehydrogenase (6-PGDH) are also involved in lipogenesis by supplying NADH, an essential factor for fatty acids and cholesterol biosynthesis. The levels of G6PDH, 6-PGDH and ME were reported to significantly increase in chronic alcohol treated rats (Choi et al., 2006).
Alterations in the NAD/NADH redox potential has been suggested to cause fatty liver due to inhibition of fatty acid oxidation and TCA cycle in addition to stimulating lipogenesis (Galli et al., 2001; Koh et al., 2004; Nagy, 2004). This process decreases the hepatic release of lipoproteins, increases the mobilization of peripheral fats, enhances the uptake of circulating lipids and decreases the fatty acid oxidation and thus increases the retention of lipids in the liver (Rukkumani et al., 2002). Ethanol treatment of rats has been reported to cause the translocation of fats from the peripheral adipose tissue to the liver (Lieber, 1991). Alcohol has also been reported to increase the biosynthesis and decrease the catabolism of fatty acids and cholesterol, resulting in their accumulation in the liver and causing hyperlipidaemia (Joseph et al., 1991). Increased cholesterol may be due to increased β-hydroxy-methyl-glutaryl CoA (HMG-CoA) reductase activity by ethanol. HMG-CoA reductase catalyses the rate limiting step in cholesterol biosynthesis (Ashakumari and Vijayammal, 1993).
Levels of triglyceride in the serum or liver increased sequel to alcohol ingestion due to several processes, including increased availability of free fatty acid L-glycerophosphate, decreased VLDL in the serum and decreased removal of triglyceride and cholesterol from serum due to diminished lipoprotein activity (Sugimoto et al., 2002; You et al., 2002). Recent data suggests that chronic ethanol intake also affects the activity of several transcription factors that coordinate expression of some genes involved in fatty acid metabolism, increasing Sterol Regulatory Element Binding Protein (SREBP), which primarily coordinates genes involved in the regulation of fatty acid synthesis and peroxisomal proliferation factor α (PPARα), which coordinates expression of genes involved in fatty acid oxidation (Nagy, 2004).
Pre-treatment with aqueous extract of Ziziphus mauritiana leaf prior to alcohol feeding significantly inhibited levels of both cholesterol and triglyceride in serum and liver compared to the group that ingested alcohol only. This shows that pretreatment with aqueous extract of Ziziphus mauritiana leaf can inhibit the onset of hyperlipidaemia and fatty liver induced by chronic alcohol feeding probably through the inhibition of lipogenic enzymes. Inhibition of lipogenic enzymes involved in hepatic lipogenesis had been reported (Choi et al., 2006).
Co-treatment of alcohol with aqueous extract of Ziziphus mauritiana leaf in rats did not show any significant change in both serum and liver cholesterol compared to those of rats that received alcohol only. However, there was a significant decrease in the level of serum triglycerides in rats co-treated with alcohol and aqueous extract of Ziziphus mauritiana leaf compared to rats administered alcohol only. This indicates that although pretreatment with aqueous extract of Ziziphus mauritiana leaf had significant inhibition on hyperlipidemia, co-treatment of alcohol with aqueous extract of Ziziphus mauritiana leaf was not so effective. It is evident that pre-treatment with aqueous extract of Ziziphus mauritiana leaf at the dosage used was more potent at protecting rats from chronic alcohol-induced hyperlipidemia than co-treatment of alcohol with aqueous extract of Ziziphus mauritiana leaf. It is possible that pre-treatment with aqueous extract of Ziziphus mauritiana leaf was able to alter the effect of chronic alcohol ingestion on the activities of some of these transcription factors that coordinate expression of genes involved in fatty acid oxidation leading to inhibition of lipogenic enzymes. Decreased levels of serum and liver cholesterol and triglyceride could be due to enhanced fatty acid oxidation and lipolysis.
In conclusion, pretreatment with aqueous extract of Ziziphus mauritiana leaf was found to inhibit hyperlipidemia and fatty liver more than co-treatment, while post-treatment enhanced the catabolism of both serum and liver cholesterol and triglyceride in chronic alcohol induced hyperlipidemia and fatty liver.
REFERENCES
- Ashakumari, L. and P.L. Vijayammal, 1993. Additive effects of alcohol and nicotine on lipid metabolism in rats. Indian Exp. Biol., 31: 270-274.
Direct Link - Abbel, L.L., B.B. Levey, B.B. Brodie and F.E. Kendall, 1952. A simplified method for the estimation of total cholesterol in serum and demonstration of its specificity. J. Biol. Chem., 195: 357-366.
Direct Link - Dahiru, D., E.T. William and M.S. Nadro, 2005. Protective effect of Ziziphus mauritiana leaf extract on carbon tetrachloride-induced liver injury. Afr. J. Biotechnol., 4: 1177-1179.
Direct Link - Morton, J.F., 1987. Indian Jujube. In: Fruits of Warm Climates, Morton, J.F. (Eds.). Mi-Ami, Florid, pp: 272-275.
Direct Link - Sugimoto, T., S. Yamashita, M. Ishigami, N. Sakai and K. Hirano et al., 2002. Decreased microsomal triglyceride transfer protein activity contributes to initiation of alcoholic liver steatosis in rats. J. Hepatol., 36: 157-162.
CrossRefPubMedDirect Link - You, M., M. Fischer, M.A. Deeg and D.W. Crabb, 2002. Ethanol induces fatty acid synthesis pathways by activation of sterol regulatory element-binging protein (SREBP). J. Biol. Chem., 277: 29342-29347.
PubMed








