Research Article
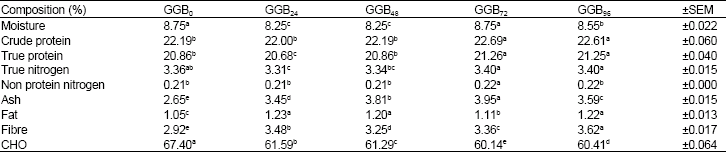
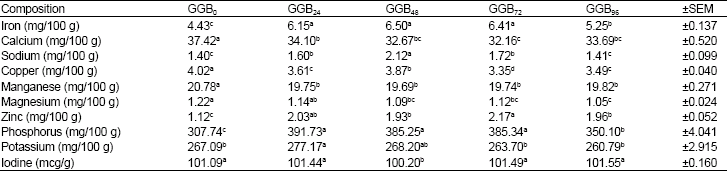
Effects of Germination on Chemical Composition of Groundbean (Kerstingiella geocarpa harm) Seeds
Department of Human Nutrition and Dietetics, Michael Okpara University of Agriculture, Umudike, PMB 7267, Umuahia, Abia State, Nigeria
I. C. Obizoba
Department of Home Science, Nutrition and Dietetics, University of Nigeria, Nsukka, Nigeria
J. U. Anyika
Department of Human Nutrition and Dietetics, Michael Okpara University of Agriculture, Umudike, PMB 7267, Umuahia, Abia State, Nigeria